

Background: Denosumab is a fully human monoclonal antibody (IgG2 subclass) that inhibits bone resorption by inhibiting RANKL, a key mediator of osteoclast formation, function, and survival. The effect of denosumab on the progression of joint damage in patients with RA on csDMARDs background treatment from the first 12 months (double-blind, placebo-controlled) of the Phase 3 DESIRABLE study has previously been reported.1
Objectives: To evaluate the safety and efficacy of long-term treatment with denosumab 60 mg every 6 months (Q6M) or every 3 months (Q3M) with RA patients in long term treatment phase of the DESIRABLE study.
Methods: Upon completion of the 12-month, double-blind phase, all patients were to receive denosumab 60 mg/Q6M or 60 mg/Q3M and calcium and vitamin D daily during the 36-month, open-label extension phase. Patients were randomized in a 1:1:2:2 ratio to receive one of four treatments (figure 1). In this analysis, the continuous administration group received 36 months of denosumab treatment, and the cross-over group received 24 months of denosumab treatment (12 month of placebo in DB phase and 24 months of denosumab treatment phase). The radiographic evaluation was the change from baseline to 12, 24 and 36 months in the van der Heijde modified total Sharp score (mTSS). Radiographs of hands and feet at baseline, 12, 24 and 36 months were scored with blinded time order by 2 readers independently. Average score of the 2 readers is used for the analysis. Missing scores were imputed using linear extrapolation/interpolation.
Figure 1 Study design

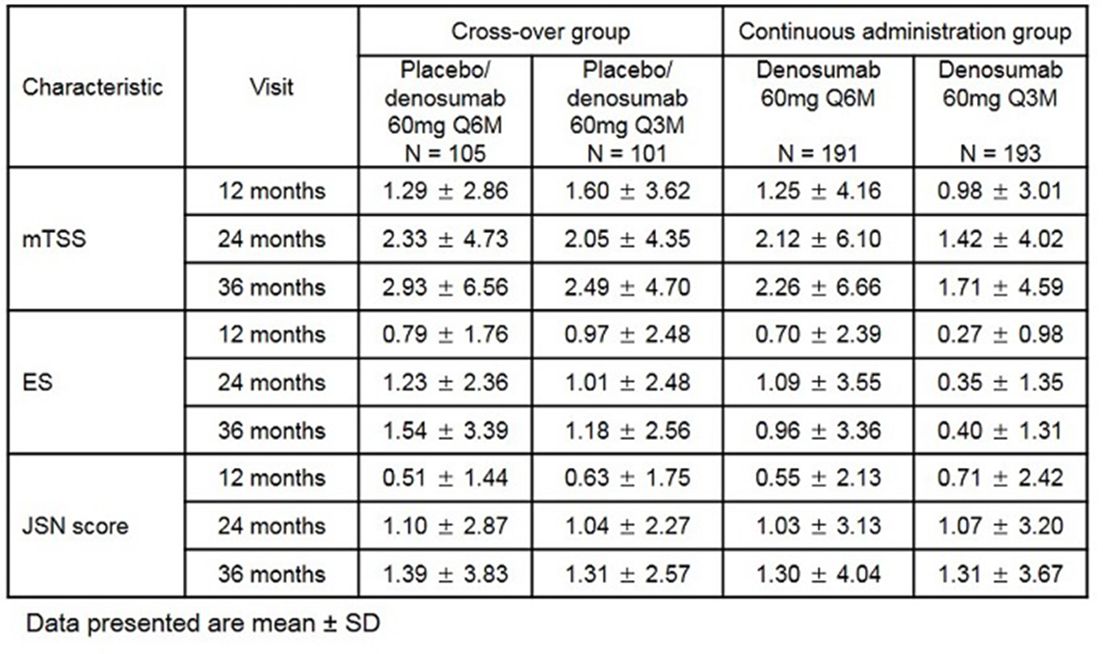
Table 1 Change from baseline in mTSS, ES and JSN score in DB and long-term treatment phase
Results: 667 (placebo/Q6M (P/Q6M), n=113; placebo/Q3M (P/Q3M), n=110; Q6M, n=222; Q3M, n=222) received at least one dose of study drug, and 607 (P/Q6M, n=105; P/Q3M, n=103; Q6M, n=199; Q3M, n=200) completed the DB period and entered the long-term extension. In both continuous administration and cross-over group, sustained inhibition of mTSS and erosion score (ES) progression was observed during long-term treatment phase (table 1). No effect on joint space narrowing (JSN) was observed (table 1). Overall proportions of patients with no progression (ie, mTSS change ≤0.5) at 36 months were 56.8% in P/Q6M, 53.3% in P/Q3M, 66.3% in Q6M group, and 65.7% in Q3M group. Incidence of adverse events (AEs), serious AEs and AEs leading to discontinuation of study drug were similar across treatment groups. No events of atypical femoral fracture were observed.
Conclusions: Denosumab treatment was associated with sustained inhibition of progression of joint destruction for up to 36 months and was generally well tolerated in Japanese patients with RA on csDMARDs. Denosumab has the potential to be a new therapeutic option to inhibit the progression of structural damage for patients with RA.
Reference:
Disclosure of Interest: T. Takeuchi Grant/research support from: Astellas, Bristol-Myers, Chugai, Daiichi Sankyo, Takeda, Teijin Pharma, AbbVie, Asahi Kasei Pharma, Mitsubishi Tanabe, Pfizer, Taisho Toyama, Eisai, AYUMI, Nipponkayaku, Consultant for: AstraZeneca, Eli Lilly, Novartis, Mitsubishi Tanabe, AbbVie, Nipponkayaku, Janssen, Astellas, Taiho, Speakers bureau: AbbVie, Bristol-Myers, Chugai, Mitsubishi Tanabe, Pfizer, Astellas, Daiichi Sankyo, Y. Tanaka Grant/research support from: Mitsubishi Tanabe, Takeda, Bristol-Myers, Chugai, Astellas, AbbVie, MSD, Daiichi Sankyo, Pfizer, Kyowa Hakko Kirin, Eisai, Ono, Speakers bureau: Daiichi Sankyo, Astellas, Pfizer, Mitsubishi Tanabe, Bristol-Myers, Chugai, YL Biologics, Eli Lilly, Sanofi, Janssen, UCB, S. Soen Grant/research support from: Chugai, Daiichi Sankyo, Speakers bureau: Asahi Kasei Pharma, Astellas, MSD, Daiichi Sankyo, Takeda, Chugai, Teijin Pharma, Pfizer, Eli Lilly Japan, Mitsubishi Tanabe, H. Yamanaka Grant/research support from: MSD, Astellas, AbbVie, BMS, Kaken, UCB, Ono, Ayumi, Eisai, Daiichi Sankyo, Takeda, Mitsubishi Tanabe, Chugai, Teijin Pharm, Torii, Nipponshinyaku, Pfizer, Consultant for: Pfizer, YL biologics, Takeda, Teijin Pharm, BMS, Nipponkayaku, Chugai, Mitsubishi Tanabe, Daiichi Sankyo, Astellas, T. Yoneda Grant/research support from: Grants-in-Aid for Scientific Research, MEXT #17H04377, Consultant for: Daiichi Sankyo, Employee of: Indiana University, S. Tanaka Consultant for: Amgen Astellas, MSD, AbbVie, Daiichi Sankyo, Eli Lilly, Ono, Asahi Kasei Pharma, Teijin Pharma, T. Nitta Employee of: Daiichi Sankyo, N. Okubo Shareholder of: Daiichi Sankyo, Employee of: Daiichi Sankyo, H. Genant Consultant for: Daiichi Sankyo, Pfizer, Amgen, Bioclinica, Eli-Lilly, Janssen, Servier, Novartis, Takeda, Merck, Biomarin, Clemencia, Agnovos, Regeneron, D. van der Heijde Consultant for: AbbVie, Amgen, Astellas, AstraZeneca, BMS, Boehringer Ingelheim, Celgene, Daiichi, Eli-Lilly, Galapagos, Gilead, Glaxo-Smith-Kline, Janssen, Merck, Novartis, Pfizer, Regeneron, Roche, Sanofi, Takeda, UCB, Employee of: Imaging Rheumatology bv.
DOI: 10.1136/annrheumdis-2018-eular.2290