

Background: Rheumatoid arthritis (RA) is a chronic inflammatory disease that causes hyperplasia of synovial tissue. Death receptor 3 (DR3) is a tumor necrosis factor receptor and binds to TL1A, a member of the TNF family. DR3 is involved in the mechanism of cell proliferation and apoptosis through NF-κB signaling. Suppression of DR3 in rheumatoid synovial fibroblasts (RA-FLS) is associated with hyperplasia of rheumatoid synovial tissue [1]. We previously revealed the expression profiles regulated by TL1A, suggesting that TL1A might affect the pathogenesis of RA, including proliferation, regulation of B cells and T cells, inflammation, and cytokine processing [2].
Objectives: In this study, we investigated the gene expression profiles regulated by DR3 in RA-FLS to reveal how DR3 is involved in the pathogenesis of RA.
Methods: RA-FLS were from patients with RA. Four individual lines of primary cultured RA-FLS were incubated either with 1000 ng/ml of human DR3-Fc protein or 1000ng/ml of human IgG1 as a control for 12h. Gene expressions were detected by microarray assay.
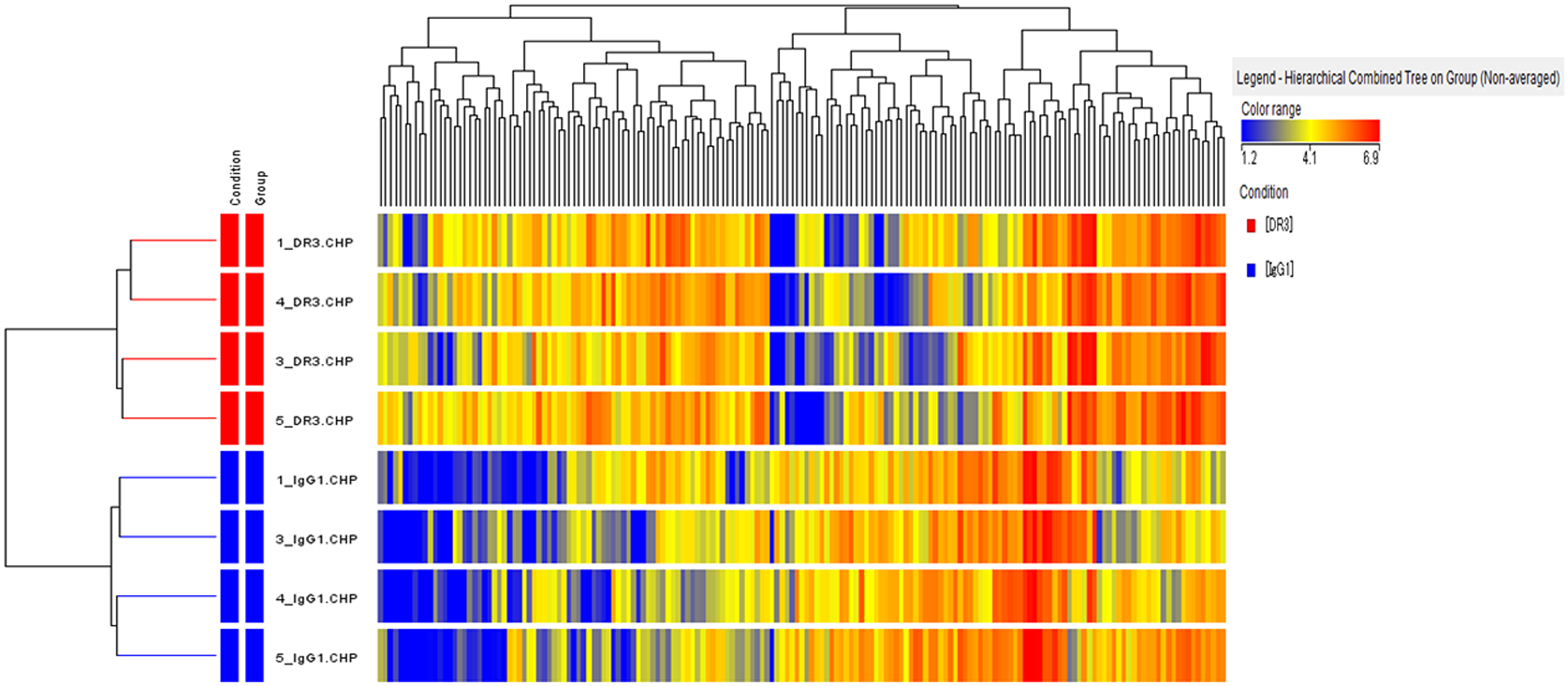
Results: Microarray data analysis revealed that DR3 up-regulated or down-regulated the expression of various genes in RA-FLS (Figure). The function of regulated genes included protein-L-isoaspartate (D-aspartate) O-methyltransferase activity, carboxyl-O-methyltransferase activity, protein carboxyl O-methyltransferase activity, regulation of cilium assembly, O-methyltransferase activity, regulation of plasma membrane bounded cell projection assembly, regulation of cell projection assembly, regulation of organelle assembly, protein methyltransferase activity, and S-adenosylmethionine-dependent methyltransferase activity. The most up-regulated 2 genes by DR3 were KIAA1109 (KIAA1109), and adhesion G protein-coupled receptor A3 (ADGRA3). The most down-regulated 2 genes by DR3 were RNA exonuclease 2 (REXO2), and family with sequence similarity 120A (FAM120A).
Conclusion: In this study, we first revealed the expression profiles of genes regulated by DR3 in RA-FLS. KIAA1109/TENR/IL2/IL21 gene is strongly associated with RA in European descent populations [3]. ADGRA3 is a member of G protein-coupled receptors (GPCRs). GPCRs associates with the regulation of cytoskeletal organization, the cell adhesion and migration, cell proliferation and apoptosis, and cell differentiation [4]. Loss of REXO2 affects cell growth and morphology [5], and REXO2 was identified as a target gene for inflammatory bowel disease-associated variants [6]. FAM120A regulates activity of Src kinase to protect cells from oxidative stress-induced apoptosis [7]. DR3 regulates the gene expressions of various key molecules in RA-FLS and may affect the pathogenesis of RA by regulating gene expression of RA-FLS.
REFERENCES:
[1]Takami N. et al., Arthritis Rheuma. 2006;54:779-787.
[2]Fukuda K. et al., Biomed Rep. 2019;1:1-5.
[3]Teixeira VH. et al., Arthritis Research & Therapy. 2009;11:R45.
[4]Hamann J. et al., Pharmacol Rev. 2015;67:338-367.
[5]Bruni F, et al., PLoS One. 2013;8:e64670.
[6]Hulur I, et al., BMC Genomics. 2015;16:138.
[7]Tanaka M. et al., Mol Cell Biol. 2009;29:402-413.
Disclosure of Interests: None declared