

Background: Patients with Rheumatoid Arthritis (RA) ≥65 years old constitute an important and not very well studied group. Even though the course of the disease may be similar to that of younger patients, treatment is usually less aggressive given the limited information on efficacy, especially of biological treatments, in this age group.
Objectives: To describe the characteristics of patients with RA ≥65 years old who started a second biological agent. To compare the survival of this second-line treatment between patients ≥65 and <65 years old.
Methods: Retrospective, observational and longitudinal study. Patients diagnosed of RA, who started a second biological agent between 2000 and 2019, who discontinued a first-line TNF inhibitor, were included. Demographic, clinical and analytical data were obtained. The sample was divided in 2 groups: <65 and ≥65 years old. Kaplan Meier and Log-rank survival analysis were performed, as well as Cox regression to identify related factors.
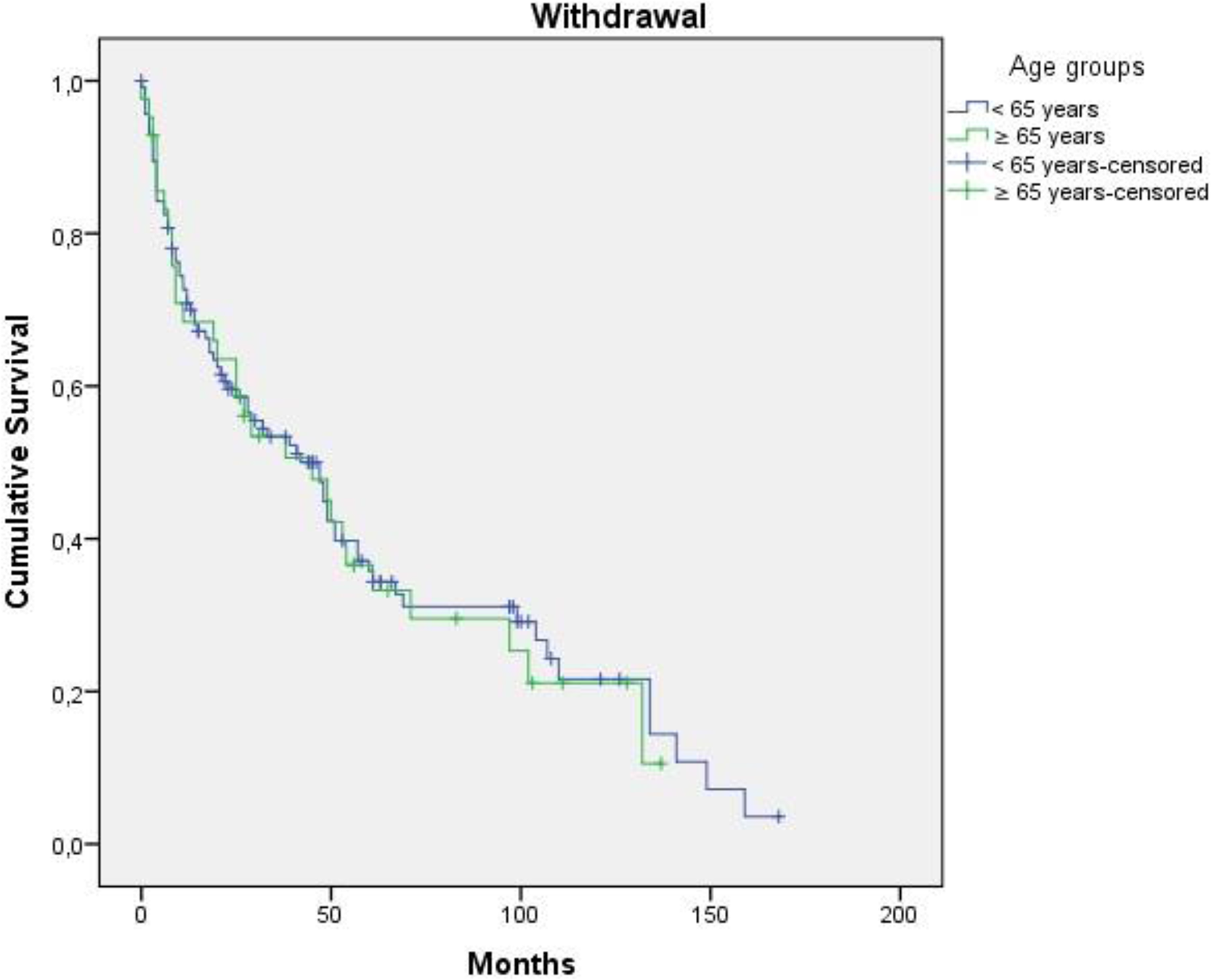
Results: 157 patients were identified, 42 (26.8%) were ≥65 years old. In this group, 73.8% were women, with a mean age at the beginning of second biological treatment of 71.43±4.76 years. Demographic and clinical data of ≥65 years old patients are shown in the table. The most frequent second biological agent was Rituximab (23.8%), followed by Adalimumab (21.4%) and Tocilizumab (19%). 76.2% of patients had a disease-modifying drug associated, being Methotrexate the most frequent (45.2%). Discontinuation of second biological agent occurred in 30 patients (71.42%) ≥65 years old, which is similar to the percentage found in patients <65 years old (66.96%; p=0.70). The main causes of withdrawal of second-line agent in patients ≥65 years were adverse effects (23.8%) and secondary failure (23.8%), whereas in <65 years were primary and secondary failure (18.3% in both). Infections were more frequent in patients ≥65 years (14.3%) in comparison with patients <65 years (6.1%). In the survival analysis of the second biological agent, patients ≥65 years presented a median survival of 45 months (IC-95%=14.10-75.90); while patients <65 years had a median survival of 47 months (IC-95%=29.55-64.46), without statistically significant differences (p=0.803) (See Figure). Among elderly patients no statistically significant differences were found after comparison of survival curves in the subgroups: 65-69, 70-74 and ≥75 years. Rituximab presented a higher survival rate in patients ≥65 years (84.3 months; p<0.001), followed by Abatacept (58.5 months). Smoking (HR=13.96; IC- 95%=2.12-91.93), erosions (HR=7.04; IC-95%=1.05-47.31) and diabetes mellitus (HR=13.37; IC-95%=1.25-143.46) were identified as risk factors for discontinuation of second biologic agent.
Conclusion: The survival of second biological agent after the failure of a first TNF inhibitor in patients ≥65 years is similar to the survival in younger patients, although there was a higher percentage of adverse effects in the first group. Rituximab and Abatacept showed a higher survival in patients ≥65 years. Smoking, erosions and diabetes mellitus were associated with an increased risk for the withdrawal of the second-line biological therapy.
REFERENCES:
[1]Richter M, Matteson E, DavisIII J, Achenbach S, Crowson C. Comparison of Biologic Discontinuation in Patients With Elderly-Onset Versus Younger-Onset Rheumatoid Arthritis. ACR Open Rheumatology. 2019; 1(10): 627–631.
| Variables | n= 42 (mean ± SD or %) |
|---|---|
| Age at diagnosis (years) | 56.48 ± 9.94 |
| Age at the beginning of the treatment (years) | 71.43 ± 4.76 |
| Women | 31 (73.8%) |
| Smokers | 11 (26.2%) |
| RF (+) | 35 (83.3%) |
| ACPA (+) | 30 (71.4%) |
| Erosions | 29 (69%) |
| Arterial hypertension | 21 (50%) |
| Diabetes mellitus | 4 (9.5%) |
| Dyslipidemia | 11 (26.2%) |
| Disease-modifying drug | |
| Methotrexate | 19 (45.2%) |
| Leflunomide | 10 (23.8%) |
| Sulfasalazine | 1 (2.4%) |
| Hydroxychloroquine | 0 (0%) |
| Infections | |
| Respiratory infection | 3 (7.1%) |
| Skin/Soft tissues infections | 2 (4.8%) |
| Herpes zoster | 1 (2.4%) |
Disclosure of Interests: None declared