

Background: EULAR has issued updated guidelines for the management of rheumatoid arthritis (RA) using conventional, biologic, and targeted synthetic DMARDs. In the 2019 update, the task force revised the preference of bDMARDs over tsDMARDs. In routine clinical practice, baricitinib is commonly used as second line or after. However, there is little information about the clinical efficacy and safety profile of baricitinib after failure of the previous agent, including another tsDMARD.
Objectives: The aim of this study was to evaluate the short-term effectiveness and safety profiles of baricitinib after insufficient response (IR) to bDMARDs or tsDMARDs in patients with RA in clinical settings.
Methods: RA patients who had been treated with baricitinib after failure of the previous agent were registered in the TBCR, a Japanese multicenter registry for RA patients treated with biologics or JAK inhibitors and followed for at least 24 weeks. Patients were divided into two groups according to the cause of failure of the previous treatment; IR (“After IR” group) and the others (“After non-IR” group). “After IR” group was further divided into four groups according to the previous agent; TNF inhibitor (TNFi group), IL-6 receptor inhibitor (IL-6Ri group), abatacept (ABT group) and tofacitinib (Tofa group). We assessed disease activities by CDAI score and drug retention rates between these groups. Furthermore, discontinuation rates due to IRs and adverse events (AEs) were evaluated.
Results: A total of 86 consecutive RA patients were registered in this study. The previous treatment was as follows; TNF inhibitor: 38 (44.2%), IL-6 receptor inhibitor: 23 (26.7%), abatacept: 11 (12.8%), tofacitinib: 13 (15.1%) and the other: 1 (1.2%).The cause of failure of the previous therapy were IRs (n=74: 86%), AEs:(n=6: 7.0%) and the others (n=6: 7.0%). In “After IR” group, the most common previous agents were TNFis (
Clinical characteristics of the BAR2mg group and the BAR4mg group
| Groups | Cases; n (%) |
|---|---|
| TNFi group | 32 (43.2) |
| IL-6R group | 21 (28.4) |
| ABT group | 11 (14.9) |
| Tofa group | 9 (12.2) |
| The oter | 1 (1.4) |
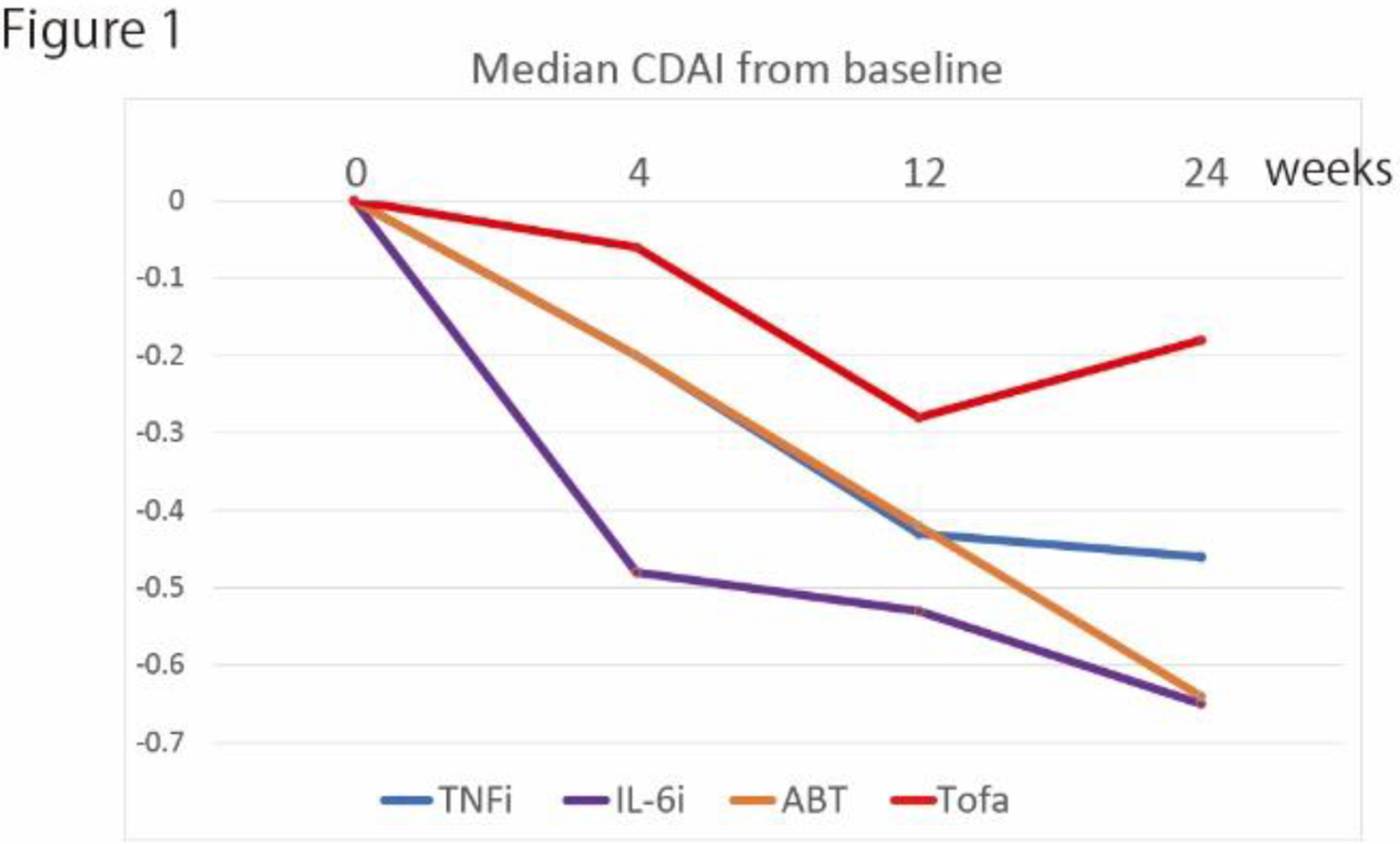
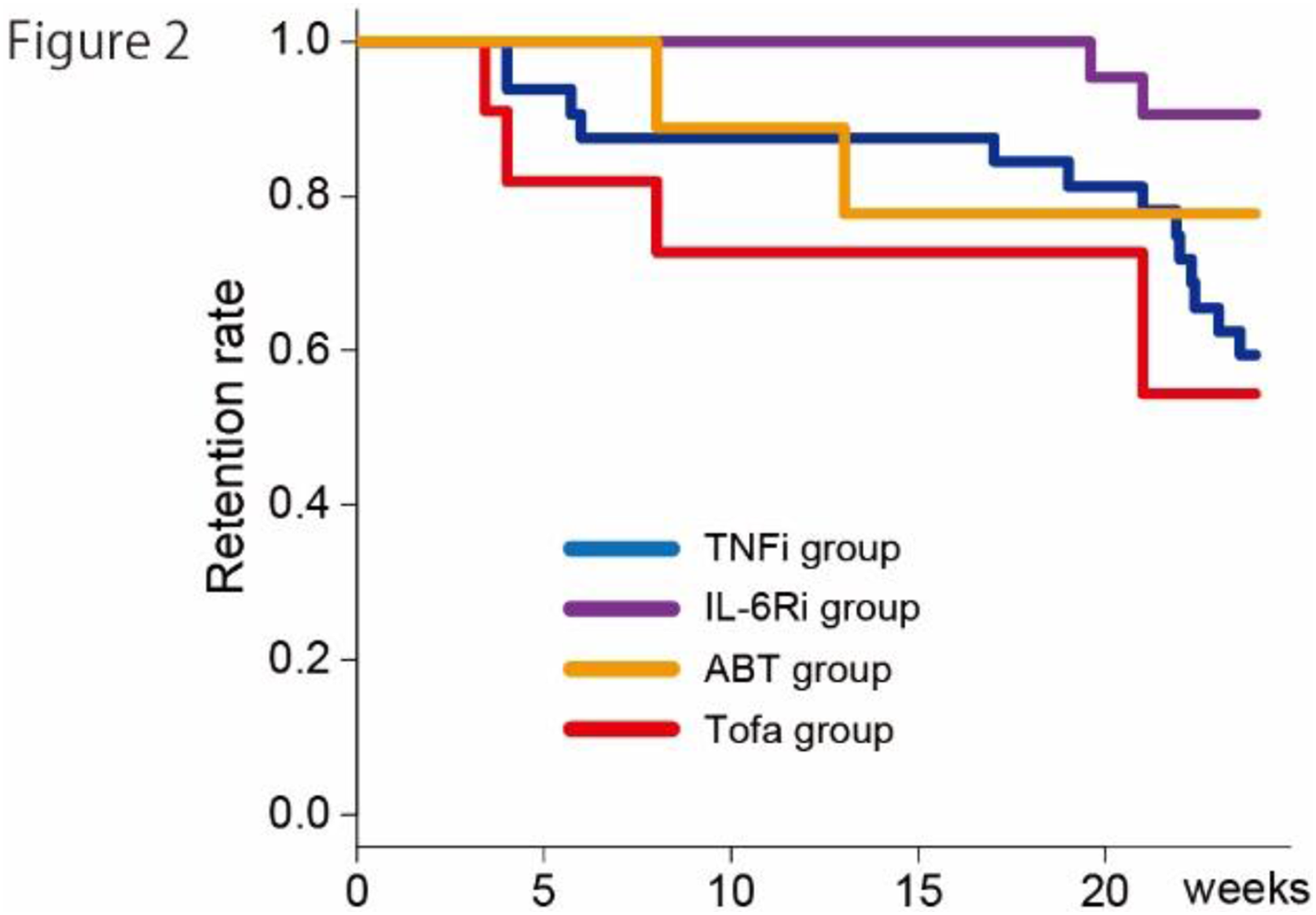
Conclusion: In this study, we demonstrated the short-term effectiveness and safety profiles of baricitinib after insufficient response to bDMARDs or tsDMARDs in patients with RA in the ‘real-world’ setting. Baricitinib improved disease activity after failure of the previous agent, even after IR to another tsDMARD. With respect to safety, the profile is almost tolerable, although careful observation is necessary for possible complications and AEs including herpes zoster.
Disclosure of Interests: Hanabayashi Masahiro Speakers bureau: Astellas Pharma Inc., Mitsubishi Tanabe Pharma Corporation, Eisai Pharma Corporation, Chugai Pharma Corporation, abbvie, Bristol-Myers Squibb, Pfizer, Janssen Pharmaceutical K.K., Eli Lilly Japan K.K. and UCB Japan, Nobunori Takahashi Speakers bureau: AbbVie, Asahi Kasei, Astellas, Bristol-Myers Squibb, Chugai, Daiichi-Sankyo, Eisai, Eli Lilly, Janssen, Mitsubishi Tanabe, Ono, Pfizer, Takeda, and UCB Japan, Toshihisa Kojima Grant/research support from: Chugai, Eli Lilly, Astellas, Abbvie, and Novartis, Consultant of: AbbVie, Speakers bureau: AbbVie, Astellas, Bristol-Myers Squibb, Chugai, Daiichi-Sankyo, Eli Lilly, Janssen, Mitsubishi Tanabe, Pfizer, and Takeda