

Background: JPsA is presented in 4 to 9% children with juvenile arthritis [1,2]. According to ILAR criteria, patients with uveitis, HLA-B27-positivity, and other exclusion criteria cannot be included in the JPsA category. Therefore, in pediatric rheumatological practice, the Vancouver criteria are more often used [3].
Objectives: to identify clinical features of psoriatic arthritis in children.
Methods: a single center, prospective and retrospective, continuous clinical trial for the period from 1989 to 2019 was conducted.
Results: We observed 83 patients with JPsA aged from 4 months to 17 years. Fifty-nine (71%) patients had definite JPsA and 24 (29%) had probable JPsA, according to the Vancouver criteria. The clinical and demographic characteristics are presented in
The clinical and demographic characteristics of patients with JPsA (n=83)
| Demographic indicators |
JPsA
|
|||
|---|---|---|---|---|
| Definite | Probable | |||
| Number of patients (n) | 59 | 24 | ||
| Girl/Boy Ratio | 1,7:1 | 2:1 | ||
| Average age, years | 6,6±4 (5) | 6,3±2,7 (5) | ||
| Duration of the disease, years | 3 ±2 (3) | |||
| Family history of psoriasis n, (%) | First-degree relative with psoriasis | 22 (26%) | ||
| Second-degree relative with psoriasis | 34 (41%) | |||
| Potential trigger n, (% ) | Infection | 22 (27%) | ||
| Trauma | 12 (14%) | |||
| Vaccination | 7 (8%) | |||
| Insolation | 5 (6%) | |||
| Stress | 3 (4%) | |||
| Not identified | 34 (41%) | |||
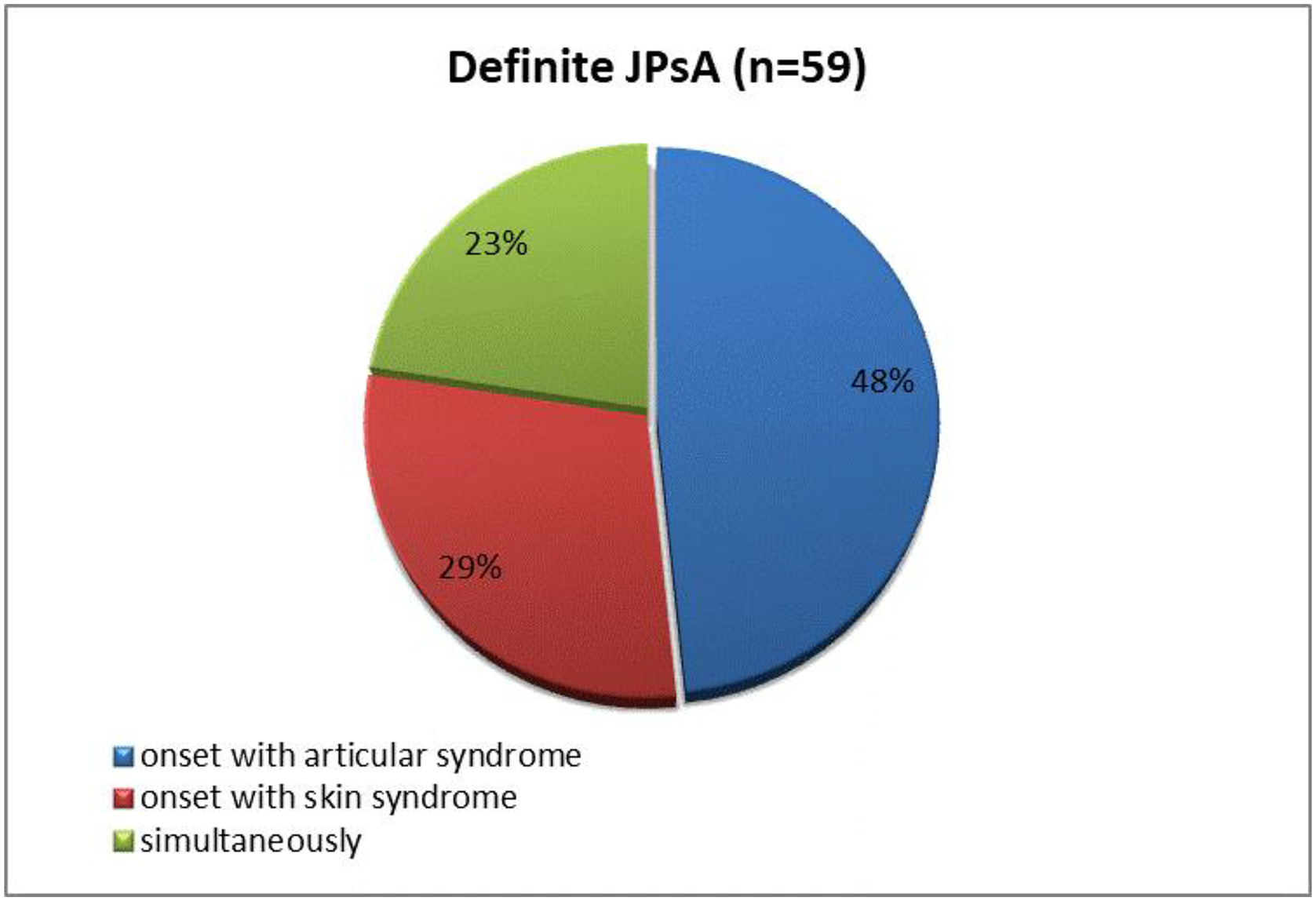
In 17 (29%) children with definite JPsA, skin lesions presented as the first sign of the disease, joint damage in these patients developed after 3.5 ± 2 (2) years. In 28 (48%) patients articular syndrome was observed on the onset with subsequent skin manifestations after 5.3 ± 3 (5) years. Fourteen (23%) children with definite JPsA had simultaneous debut of skin and articular syndromes (
Skin and / or articular syndromes in the onset of the disease in children with definite JPsA
Among 59 patients with definite JPsA, vulgar psoriasis was observed in 45 (76%) patients, guttate psoriasis in 9 (15%), isolated nail psoriasis in 3 (5%), and palmoplantar psoriasis in 2 (4%). Fifteen (28%) patients had a combination of cutaneous psoriasis with damage to the nail plates.
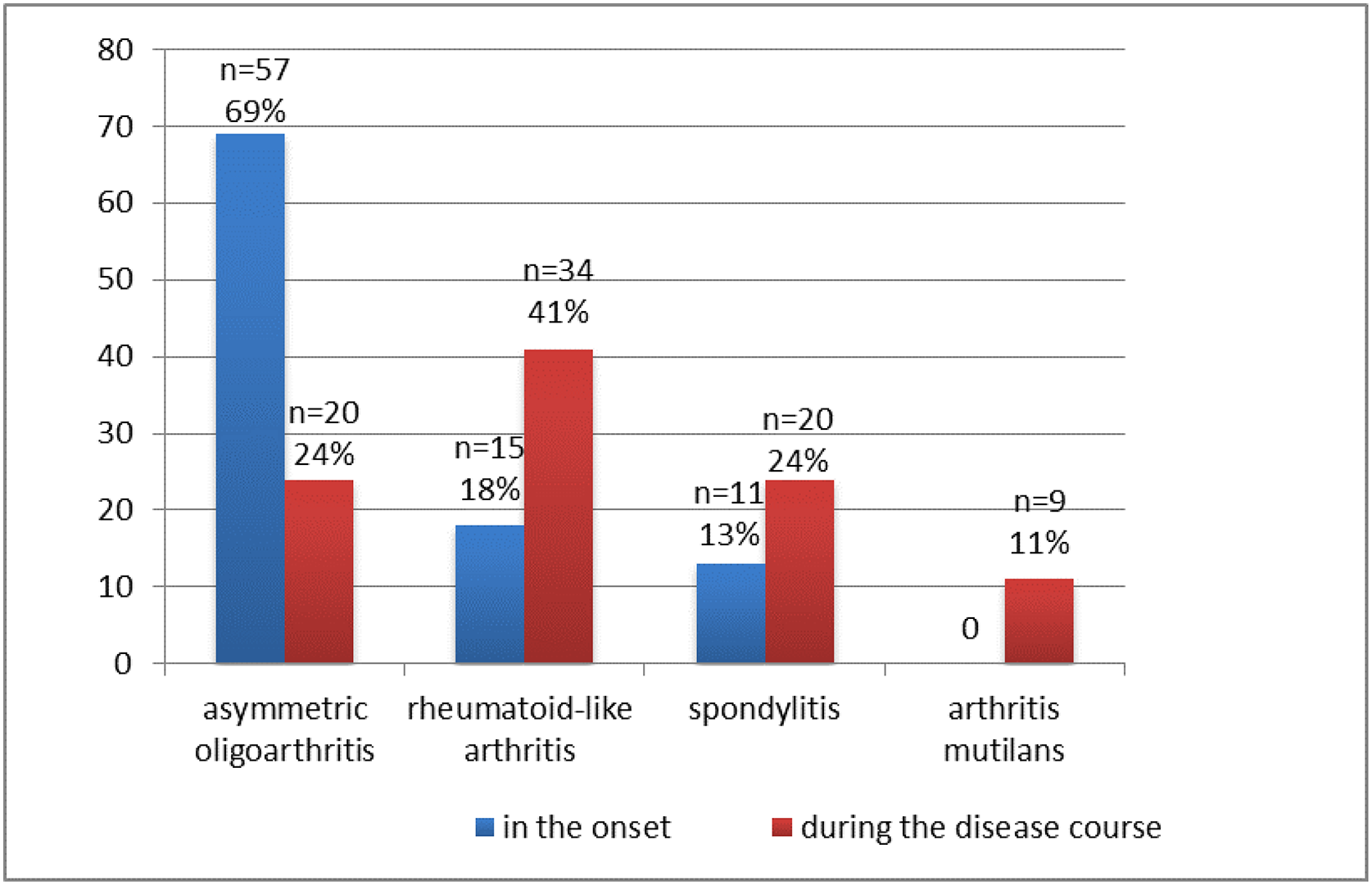
Articular syndrome in the onset of the disease was represented by oligoarticular arthritis in 57 (69%) patients, in 15 (18%) children - symmetric rheumatoid-like arthritis, and in 11 (13%) - spondylitis. The most commonly involved joints at both presentation and during the course of the disease were the knee (41%), ankle (31%), and small joints of the hands (29%). During course of the disease, the articular syndrome transformed with symmetric rheumatoid-like arthritis prevalence (
Articular syndrome JPsA in the onset and during the disease course (n, %)
In 39 (47%) children, the disease at onset was characterized by high level of ESR – 34±12 (32) mm/h.
Conclusion: In most patients with JPsA, the onset of the disease occurred at the age of 6.6±4 years. Articular syndrome in the onset of the disease was presented as oligoarticular arthritis in 69% children. Overall, transformation to symmetric rheumatoid-like arthritis was the most common. Skin lesions were represented by vulgar psoriasis in 76%, guttate psoriasis in 15%, isolated nail psoriasis in 5%, palmoplantar psoriasis in 4%, and 28% of patients had a combination of cutaneous psoriasis with damage to the nail plates.
REFERENCES:
[1]Beukelman, T., et al. The new Childhood Arthritis and Rheumatology Research Alliance (CARRA) registry: design, rationale, and characteristics of patients enrolled in the first 12 months. Pediatr Rheumatol Online J. 2017;15(1):30
[2]Horneff G., et al. Update on malignancies in children with juvenile idiopathic arthritis in the German BIKER Registry. Clin. Exp. Rheumatol. 2016; 34(6):1113–1120.
[3]Southwood T.R., et al. Psoriatic arthritis in children. Arthritis Rheum.1989; 32(8):1007-1013.
Disclosure of Interests: Karina Aleksanyan: None declared, Svetlana Chebysheva: None declared, Angelina Meleshkina: None declared, Ekaterina Popova: None declared, Elena Zholobova Grant/research support from: Novartis and Pfizer Inc, Speakers bureau: AbbVie, Novartis, Pfizer Inc and Roche