

Background: Patients with psoriatic arthritis (PsA) present with heterogeneous clinical phenotypes, which includes different clinical manifestations such as peripheral arthritis, axial disease, dactylitis, enthesitis, and skin and nail psoriasis. Many drugs for treatment of PsA are available for individualised treatment strategies but robust evidence is available for peripheral arthritis only as primary endpoint of RCTs.
Objectives: To evaluate perception of German physicians on the value of current PsA treatments on different clinical PsA manifestations.
Methods: In a face-to-face meeting, 8 German physicians (dermatology, rheumatology), specialised in PsA research/patient care, proposed initial scores for the effect size of current PsA drugs on different PsA manifestations based on knowledge of study data and personal experience in use of the drugs. The ability to achieve a consensus of the proposed efficacy scores was explored by applying an online survey among a cohort of PsA experienced physicians. Finally, a second online survey evaluated how a larger group of physicians personally estimate the drug effect sizes on different PsA manifestations.
Results:
Efficacy scores proposed by a PsA expert group and consensus achieved by an online survey among PsA treating physicians Efficacy scores for different drugs and manifestations range from 0 (no effect) to 5 (maximal effect). Green fields indicate, that at least 65% of the survey participants agreed on the proposed efficacy score.
| Peripheral arthritis | Axial disease | Enthesitis | Dactylitis | Skin disease | Nail disease | |
|---|---|---|---|---|---|---|
| Methotrexat | 2.5 | 0 | 0 | 1 | 2.5 | 1.5 |
| Sulfasalazin | 1 | 0 | 0 | 0 | 0 | 0 |
| Leflunomid | 2.5 | 0 | 0 | 1 | 1 | 1 |
| Apremilast | 3 | 1 | 3 | 2.5 | 3 | 3 |
| Etanercept | 4 | 4 | 4 | 3.5 | 3 | 3.5 |
| Infliximab | 4 | 4 | 4 | 4 | 4 | 4 |
| Adalimumab | 4 | 4 | 4 | 4 | 4 | 4.5 |
| Golimumab | 4 | 4 | 4 | 4 | 3 | 3 |
| Certolizumab pegol | 4 | 4 | 4 | 4 | 4 | 4 |
| Ustekinumab | 3.5 | 2.5 | 4.5 | 3 | 4.5 | 4 |
| Secukinumab | 4 | 4 | 4 | 4 | 5 | 4.5 |
| Abatacept | 3.5 | 0 | 0 | 0 | 1.5 | 0 |
Results from an online survey among physicians querying their perception of efficacies of PsA drugs on different manifestations. Therapeutic effectiveness is estimated by the participants on a scale from 0 (no effect) to 5 (maximal effectiveness); NA = no answer. The size of the dots relates to the number of answers given. Red stars indicate the scores proposed by the initial expert group.
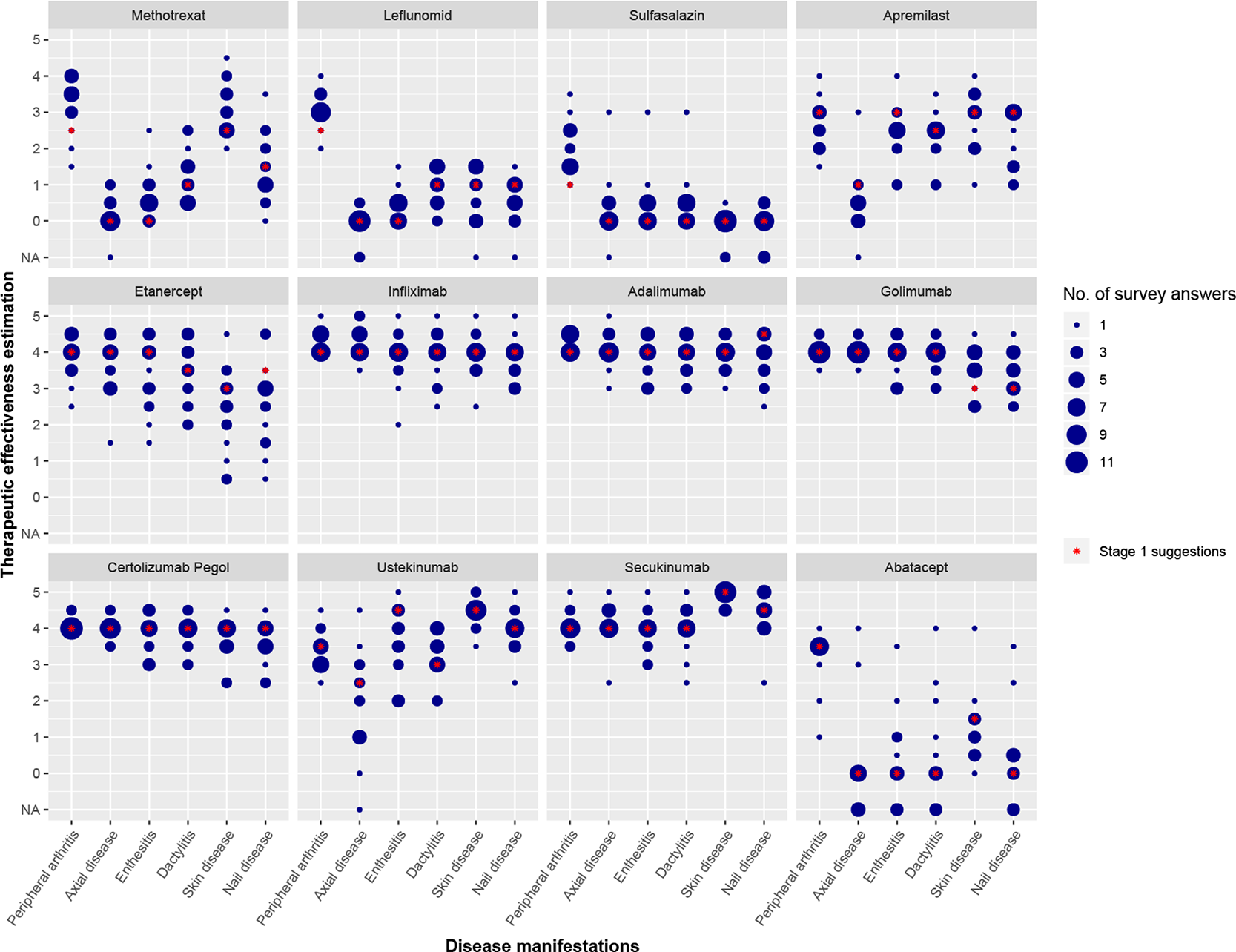
Conclusion: Many treatments are available to be used in PsA. The usage of treatment classes seems to depend on experience level and mode of action. Higher agreement on efficacy in specific diseases manifestation was achieved in the class of biologics whereas the opinion on efficacy of csDMARDs in general is divergent. Strategies are needed to guide physicians with lower level of experience in the use of specific drugs to achieve an optimised patient care level using personalized treatment strategies.
Disclosure of Interests: Timm Oberwahrenbrock Grant/research support from: BMS, Hannah Tian: None declared, Diamant Thaçi Grant/research support from: Janssen Research & Development, LLC, Klaus Krueger Consultant of: Celgene, Jürgen Wollenhaupt Consultant of: Celgene, Michaela Köhm Grant/research support from: Pfizer, Janssen, BMS, LEO, Consultant of: BMS, Pfizer, Speakers bureau: Pfizer, BMS, Janssen, Novartis, Frank Behrens Grant/research support from: Pfizer, Janssen, Chugai, Celgene, Lilly and Roche, Consultant of: Pfizer, AbbVie, Sanofi, Lilly, Novartis, Genzyme, Boehringer, Janssen, MSD, Celgene, Roche and Chugai