

Background: Systemic sclerosis (SSc) is one of the connective tissue diseases with the poorer prognosis and disease-related causes, particularly pulmonary fibrosis, PAH and cardiac involvement, accounting the most deaths.
Objectives: This multicentre study aimed to evaluate the global survival and any predictor of mortality in a large multicentric cohort of SSc patients.
Methods: We performed a retrospective analysis examining the medical records of our longitudinal SSc cohorts with a median (IQR) follow-up of 11 (6-18) years from 3 Scleroderma Units since January 2009. All clinical, laboratory and instrumental findings have been recorded and analyzed using Chi-squared tests, Kaplan-Meier curves, log-rank tests, and Cox proportional hazards models.
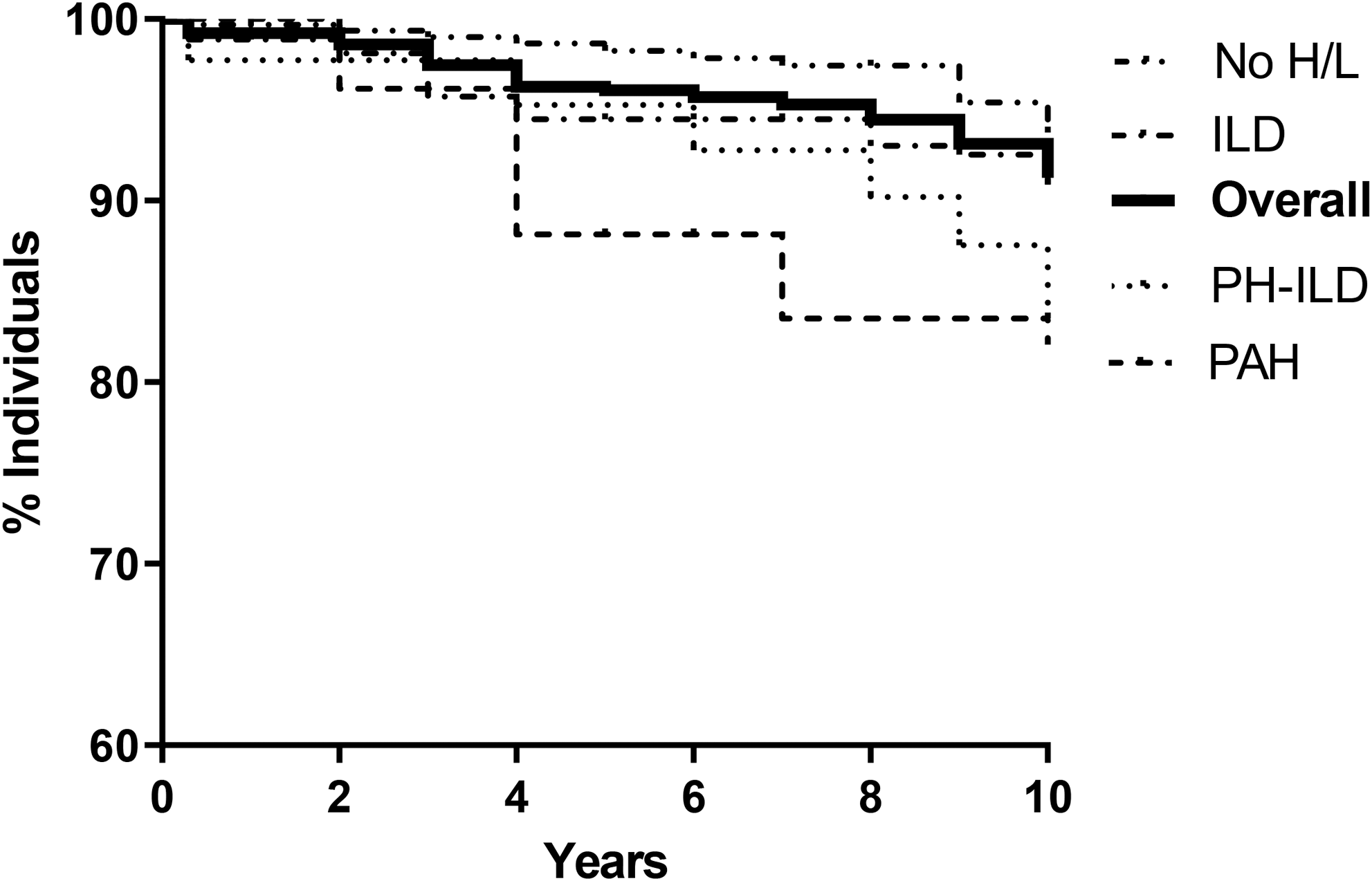
Results: Data from 750 SSc patients (91.9% female; mean (SD) age at first Non-Raynaud symptom 48.4 (15.3) years, median (IQR) disease duration 3 (0-8) years; diffuse cutaneous involvement 162 (21.6%) patients) fulfilling the 1980 ARA and/or 2013 ACR/EULAR classification criteria, were collected. All patients were positive for ANA, anti-Topo-I Abs were found in 235 (31.3%) and Cenp-B Abs in 300 (40%) patients. 98 (13.1%) patients were positive to other Abs (Anti-RNA polymerase III, anti-Pm/Scl) and anti-ENA were negative/unknown for 117 (15.6%) patients. Interstitial lung disease (ILD) was present in 202 (26.9%), pulmonary arterial hypertension (PAH) was found in 29 (3.9%), and 50/750 (6.7%) patients presented pulmonary hypertension combined with ILD (PH-ILD). The overall 10-years survival was 93.1% and, it was significantly impaired by the presence of ILD, PAH or PH-ILD [Figure]. The univariate analysis showed that female gender, higher age at first Non-Raynaud symptom, earlier referral to a tertiary Scleroderma center, absence of any ENA antibodies, and PH-ILD presence were survival predictors. After multivariate analysis the significance of PH-ILD was lost [Table]. Disease duration, basal Rodnan skin score, smoking, renal or gastrointestinal comorbidities, NYHA functional class, steroid or immune-suppressive treatments did not reach the statistically significance.
Conclusion: Our study demonstrated a global 10-years survival rate over 93%. Male patients and rapid evolution of Non-Raynaud symptoms represent the main death predictors in our SSc cohort. A rapid referral to a tertiary rheumatological centre and early treatment with effective agents are associated to a better prognosis.
Kaplan-Meier curves for 5-years survival in SSc patients (Log-rank 8.96, p=0.03).
Prognostic factors for 10-years survival at univariate and multivariate analysis.
| UNIVARIATE ANALYSIS | MULTIVARIATE ANALYSIS | |||||
|---|---|---|---|---|---|---|
| HR | 95%IC | P | HR | 95%IC | P | |
| Female gender | 0.35 | 0.15-0.81 | 0.01 | 0.31 | 0.15-0.66 | 0.002 |
| Age at first Non-Raynaud symptom | 1.07 | 1.04-1.1 | 0.001 | 1.08 | 1.05-1.11 | 0.001 |
| Time referral to a tertiary SSc centre | 0.83 | 0.76-0.92 | 0.001 | 0.84 | 0.77-0.93 | 0.001 |
| Absence of any ENA antibodies | 0.08 | 0.01-0.62 | 0.01 | 0.09 | 0.01-0.71 | 0.02 |
| PH-ILD presence | 2.6 | 1.01-6.82 | 0.04 | 2.4 | 0.93-6.1 | 0.069 |
Disclosure of Interests: Fabio Cacciapaglia Speakers bureau: BMS; Roche; Pfizer; Abbvie, Enrico De Lorenzis: None declared, Addolorata Corrado: None declared, Silvia Laura Bosello Speakers bureau: Abbvie, Pfizer, Boehringer, Marco Fornaro: None declared, Fabio Montini: None declared, Livio Urso: None declared, Lucrezia Verardi: None declared, Alberto Altomare: None declared, Francesco Paolo Cantatore: None declared, Elisa Gremese Consultant of: AbbVie, Bristol-Myers Squibb, Celgene, Eli Lilly, Janssen, Merck Sharp & Dohme, Novartis, Sanofi, UCB, Roche, Pfizer, Speakers bureau: AbbVie, Bristol-Myers Squibb, Celgene, Eli Lilly, Janssen, Merck Sharp & Dohme, Novartis, Sanofi, UCB, Roche, Pfizer, Florenzo Iannone Consultant of: Speaker and consulting fees from AbbVie, Eli Lilly, Novartis, Pfizer, Roche, Sanofi, UCB, MSD, Speakers bureau: Speaker and consulting fees from AbbVie, Eli Lilly, Novartis, Pfizer, Roche, Sanofi, UCB, MSD