

Background: Osteoarthritis (OA) is a degenerative, chronic, and progressive joint disease. It is associated with chronic pain, joint function impairments and disabilities, causing a poorer quality of life with physical and/or mental co-morbidity. Along with population ageing and increasing obesity, the incidence of OA is rising and there is an urgent need for new treatment options.
Objectives: JTA-004 is a novel protein solution in development for the treatment of knee OA pain. Supplemented with hyaluronic acid and clonidine, it is designed to provide a fast-acting and long-lasting pain relief. To evaluate efficacy and safety and to select the most effective formulation, single intra-articular administration of 3 JTA-004 formulations were tested and compared to Hylan G-F 20 during a 6-month period.
Methods: In this prospective, multicentre, double-blind phase II/III trial (NCT02740231), eligible participants were 50-79-year-old men and women with primary knee OA classified with Kellgren-Lawrence grade II or III and a body mass index (BMI) under 35. 164 patients were randomly assigned to one of the three JTA-004 formulations or the reference treatment (Hylan G-F 20) in a 1:1:1:1 ratio. The three JTA-004 formulations differed in their clonidine concentration (50 or 100 µg/ml) and/or their volume of injection (2 or 4 ml) (
Description of the three JTA-004 formulations administered
| JTA-004 formulation | Plasma protein
| Clonidine | HA | Volume of | |||
|---|---|---|---|---|---|---|---|
| Concentration | Amount | Concentration | Amount | Concentration | Amount | injection | |
| JTA-100/2 | 1.02 g/ml | 2.04 g | 50 µg/ml | 100 µg | 10 mg/ml | 20 mg | 2 ml |
| JTA-200/2 | 1.02 g/ml | 2.04 g | 100 µg/ml | 200 µg | 10 mg/ml | 20 mg | 2 ml |
| JTA-200/4 | 1.02 g/ml | 4.08 g | 50 µg/ml | 200 µg | 10 mg/ml | 40 mg | 4 ml |
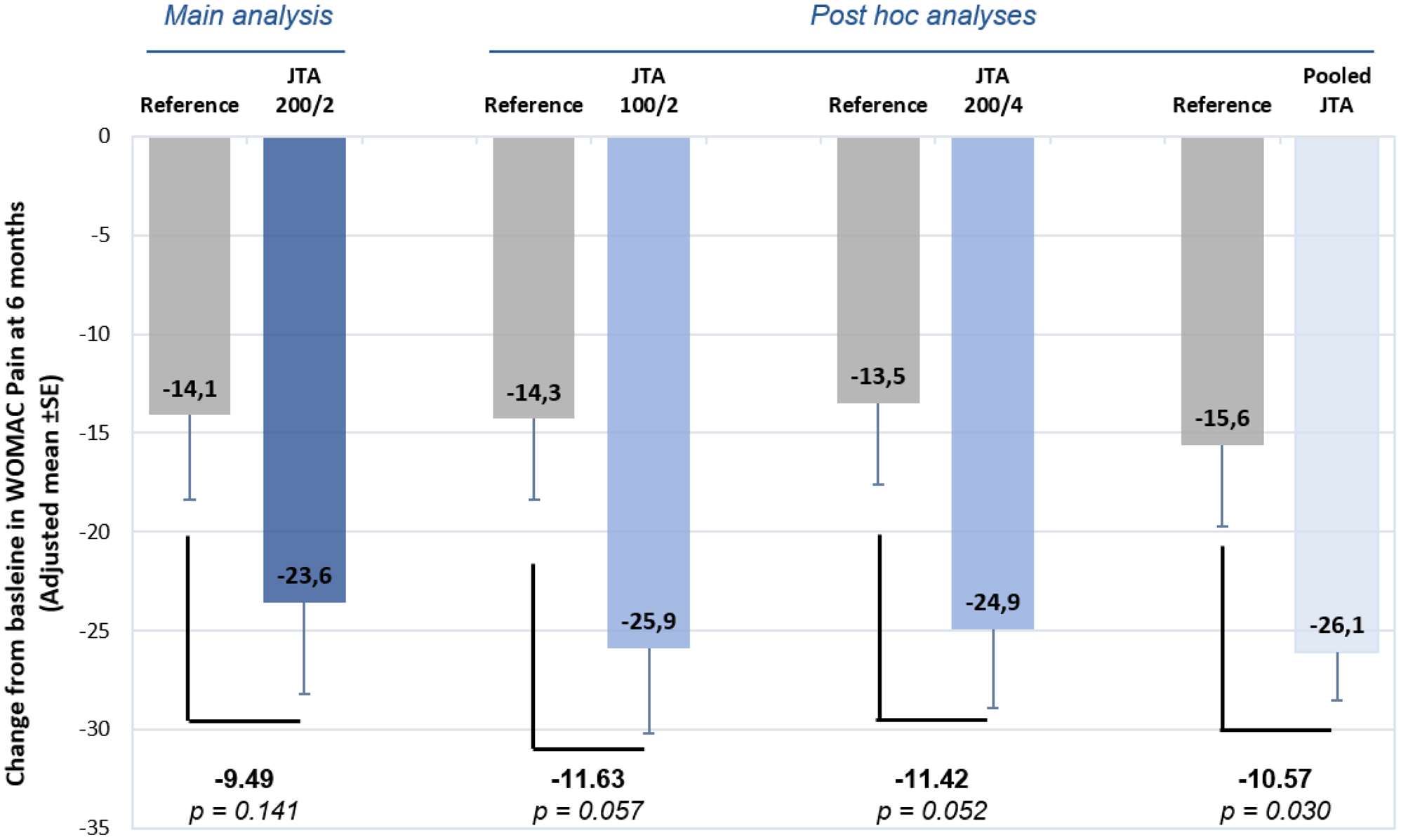
Results: At 6 months, patients in the three JTA-004 groups showed a better improvement in pain compared to patients in the reference group. The between-group difference (between each JTA-004 test group and reference group) in adjusted (adapted to difference in baseline values) mean change in WOMAC
®
Pain Subscale Score from baseline ranged between -9.49 mm and -11.63 mm at 6 months post-injection. Statistical superiority of each JTA-004 formulation over Hylan G-F 20 was however not demonstrated (p-value between 0.052 and 0.141) (
Main and post hoc analyses.
All JTA-004 formulations were shown to be well tolerated and had a clinically acceptable safety profile. There was a trend for fewer treatment-related events in the JTA-100/2 group, notably no cases of post-injection mild and transient hypotension.
Conclusion: This study provides a first evidence of efficacy and safety of JTA-004 in the treatment of knee OA pain.
Disclosure of Interests: Marie Bettonville Employee of: Bone Therapeutics, Marc Leon: None declared, Joëlle Margaux: None declared, Didier Urbin-Choffray: None declared, Emilie Theunissen: None declared, Tatiana Besse-Hammer: None declared, Yves Fortems: None declared, Séverine Verlinden: None declared, Olivier Godeaux Consultant of: Bone Therapeutics, Anne-Sophie Delmarcelle Employee of: Bone Therapeutics, Jean-François Kaux Consultant of: Bone Therapeutics