

Background: The Phase IIIb A ssessing V ery E arly R A T reatment (AVERT)-2 trial (NCT02504268) evaluated SC abatacept (ABA) + MTX vs ABA placebo (PBO) + MTX in ACPA+ patients (pts) with early, active RA. 1 Results from the 56-wk induction period (IP) showed a significantly greater proportion of pts treated with ABA + MTX (vs MTX alone) reported clinically meaningful improvements in HAQ-DI, global disease activity and pain, which were sustained at 52 wks. 2
Objectives: To report maintenance of SDAI remission and PROs from the AVERT-2 de-escalation (D-E) period.
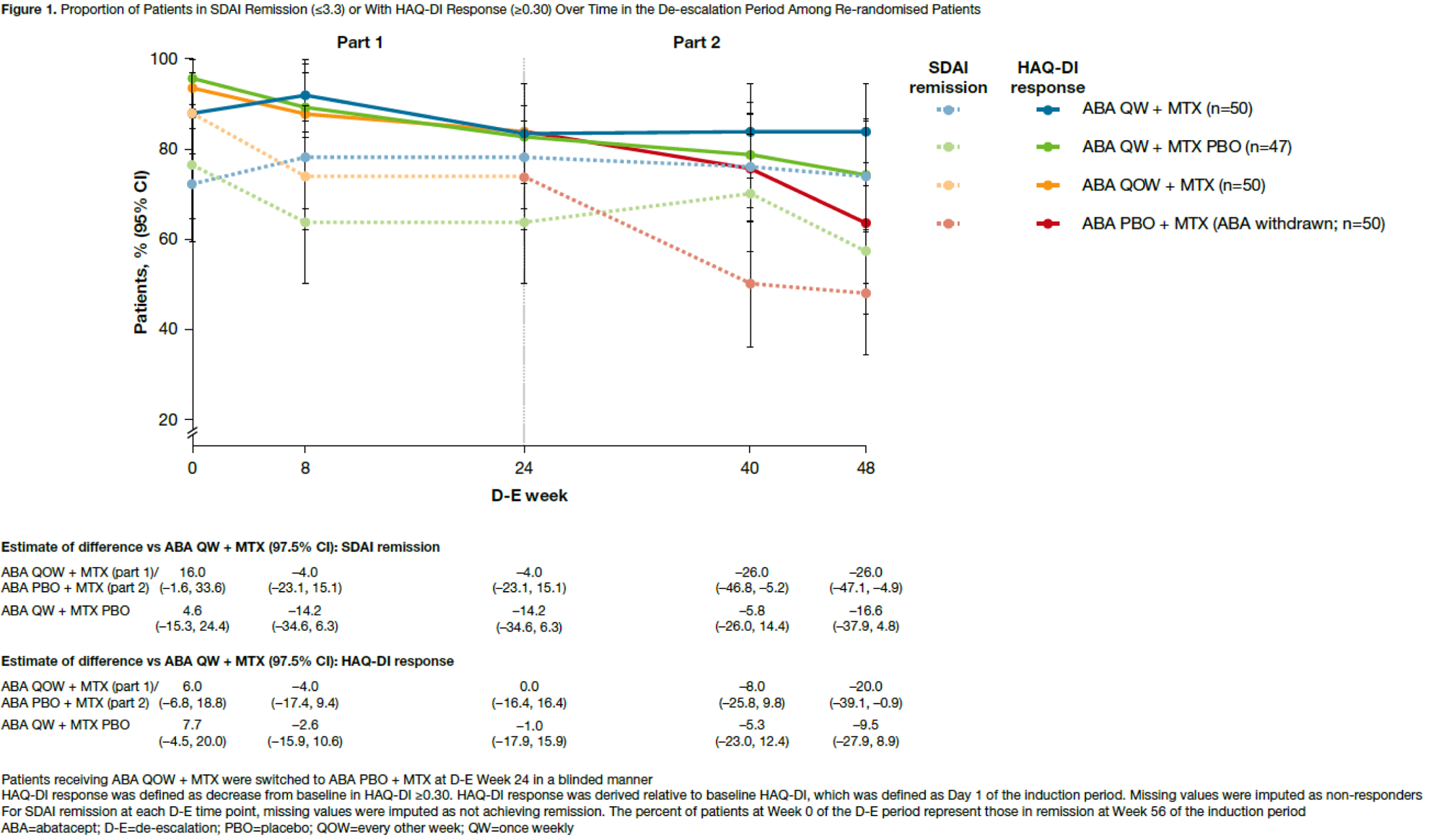
Methods: Pts received blinded SC ABA (125 mg once wkly [QW]) + MTX or ABA PBO + MTX induction treatment for 56 wks. In this analysis, pts who completed induction with ABA + MTX and had sustained SDAI remission (≤3.3 at Wks 40 and 52) were re-randomised 1:1:1 to ABA QW + MTX, stepwise D-E (ABA every other wk + MTX for 24 wks then ABA PBO + MTX for 24 wks), or ABA QW + MTX PBO for 48 wks in the D-E period. PROs included physical function (HAQ-DI [0–3; decrease=improvement] and Short-Form 36 [SF-36] v2.0 Physical Functioning Scale [PFS]; 0–100; increase=improvement), and fatigue (Functional Assessment of Chronic Illness Therapy-Fatigue [FACIT-F] score; 0–52; decrease=improvement). Endpoints included: proportion of pts in SDAI remission and pts with HAQ-DI response (decrease from IP Day [D]1 in HAQ-DI ≥0.30); adjusted mean change (adMC) from D-E D1 in HAQ-DI, SF-36 PFS or FACIT-F to D-E Wk 48. adMCs were estimated using a mixed effect model with repeated measures.
Results: 147 ABA + MTX-treated pts were re-randomised in the D-E period. Across re-randomised arms, the range of mean scores was 1.87–2.52 for SDAI and 0.18–0.30 for HAQ-DI at entry into D-E period (D-E D1). 74% of pts receiving ABA QW + MTX maintained SDAI remission at D-E Wk 48 (
Conclusion: In the AVERT-2 D-E period, continued combination therapy (abatacept + MTX) resulted in maintenance of benefits on PROs, particularly physical functioning, in seropositive pts with early RA. D-E of abatacept followed by complete withdrawal was associated with the greatest loss of remission as well as worsening of PROs. The PRO results corresponded well to the maintenance of clinical (SDAI) remission.
REFERENCES:
[1]Emery P, et al. ACR 2018; San Diego, USA: Poster 563.
[2]Emery P, et al. ACR 2019; Atlanta, USA: Poster 1423.
Acknowledgments: Joanna Wright (medical writing, Caudex; funding: Bristol-Myers Squibb)
Disclosure of Interests: Paul Emery Grant/research support from: AbbVie, Bristol-Myers Squibb, Merck Sharp & Dohme, Pfizer, Roche (all paid to employer), Consultant of: AbbVie (consultant, clinical trials, advisor), Bristol-Myers Squibb (consultant, clinical trials, advisor), Lilly (clinical trials, advisor), Merck Sharp & Dohme (consultant, clinical trials, advisor), Novartis (consultant, clinical trials, advisor), Pfizer (consultant, clinical trials, advisor), Roche (consultant, clinical trials, advisor), Samsung (clinical trials, advisor), Sandoz (clinical trials, advisor), UCB (consultant, clinical trials, advisor), Yoshiya Tanaka Grant/research support from: Asahi-kasei, Astellas, Mitsubishi-Tanabe, Chugai, Takeda, Sanofi, Bristol-Myers, UCB, Daiichi-Sankyo, Eisai, Pfizer, and Ono, Consultant of: Abbvie, Astellas, Bristol-Myers Squibb, Eli Lilly, Pfizer, Speakers bureau: Daiichi-Sankyo, Astellas, Chugai, Eli Lilly, Pfizer, AbbVie, YL Biologics, Bristol-Myers, Takeda, Mitsubishi-Tanabe, Novartis, Eisai, Janssen, Sanofi, UCB, and Teijin, Vivian Bykerk: None declared, Thomas Huizinga Grant/research support from: Ablynx, Bristol-Myers Squibb, Roche, Sanofi, Consultant of: Ablynx, Bristol-Myers Squibb, Roche, Sanofi, Gustavo Citera Grant/research support from: AbbVie, Amgen, Eli Lilly, Gema, Genzyme, Novartis and Pfizer Inc, Consultant of: AbbVie, Amgen, Eli Lilly, Gema, Genzyme, Novartis and Pfizer Inc, Clifton Bingham Grant/research support from: Bristol-Myers Squibb, Consultant of: Bristol-Myers Squibb, Subhashis Banerjee Shareholder of: AbbVie, Bristol-Myers Squibb, Lily, Pfizer, Employee of: Bristol-Myers Squibb (current); AbbVie, Lily, Pfizer (past), Sean Connolly Shareholder of: Bristol-Myers Squibb, Employee of: Bristol-Myers Squibb, Joe Zhuo Shareholder of: Bristol-Myers Squibb, Employee of: Bristol-Myers Squibb, Robert Wong Shareholder of: Bristol-Myers Squibb, Employee of: Bristol-Myers Squibb, Kuan-Hsiang Gary Huang Shareholder of: Bristol-Myers Squibb, Employee of: Bristol-Myers Squibb, Karissa Lozenski Employee of: Bristol-Myers Squibb, Yedid Elbez Consultant of: Bristol-Myers Squibb, Roy Fleischmann Grant/research support from: AbbVie, Akros, Amgen, AstraZeneca, Bristol-Myers Squibb, Boehringer, IngelhCentrexion, Eli Lilly, EMD Serono, Genentech, Gilead, Janssen, Merck, Nektar, Novartis, Pfizer, Regeneron Pharmaceuticals, Inc., Roche, Samsung, Sandoz, Sanofi Genzyme, Selecta, Taiho, UCB, Consultant of: AbbVie, ACEA, Amgen, Bristol-Myers Squibb, Eli Lilly, Gilead, GlaxoSmithKline, Novartis, Pfizer, Sanofi Genzyme, UCB