

Background: Rheumatoid arthritis (RA) patients treated with advanced therapy (biologic disease-modifying antirheumatic drugs and targeted synthetic disease-modifying antirheumatic drugs) may be considered dose tapering after reaching treatment goal. 1 In EULAR 2016 recommendations, dose reduction can be considered if patients reach sustained remission. 2 A dose tapering policy of advanced therapy was introduced in the treatment guideline of RA since 2014 under the National Health Insurance (NHI) in Taiwan. The new reimbursement policy requests the dosage to be tapered in patients who received advanced therapy for 2 years and reached low disease activity defined by DAS28 (ESR).
Objectives: This retrospective study aims to investigate the impact of dose tapering policy on prescription pattern of advanced therapy for RA patients in Taiwan.
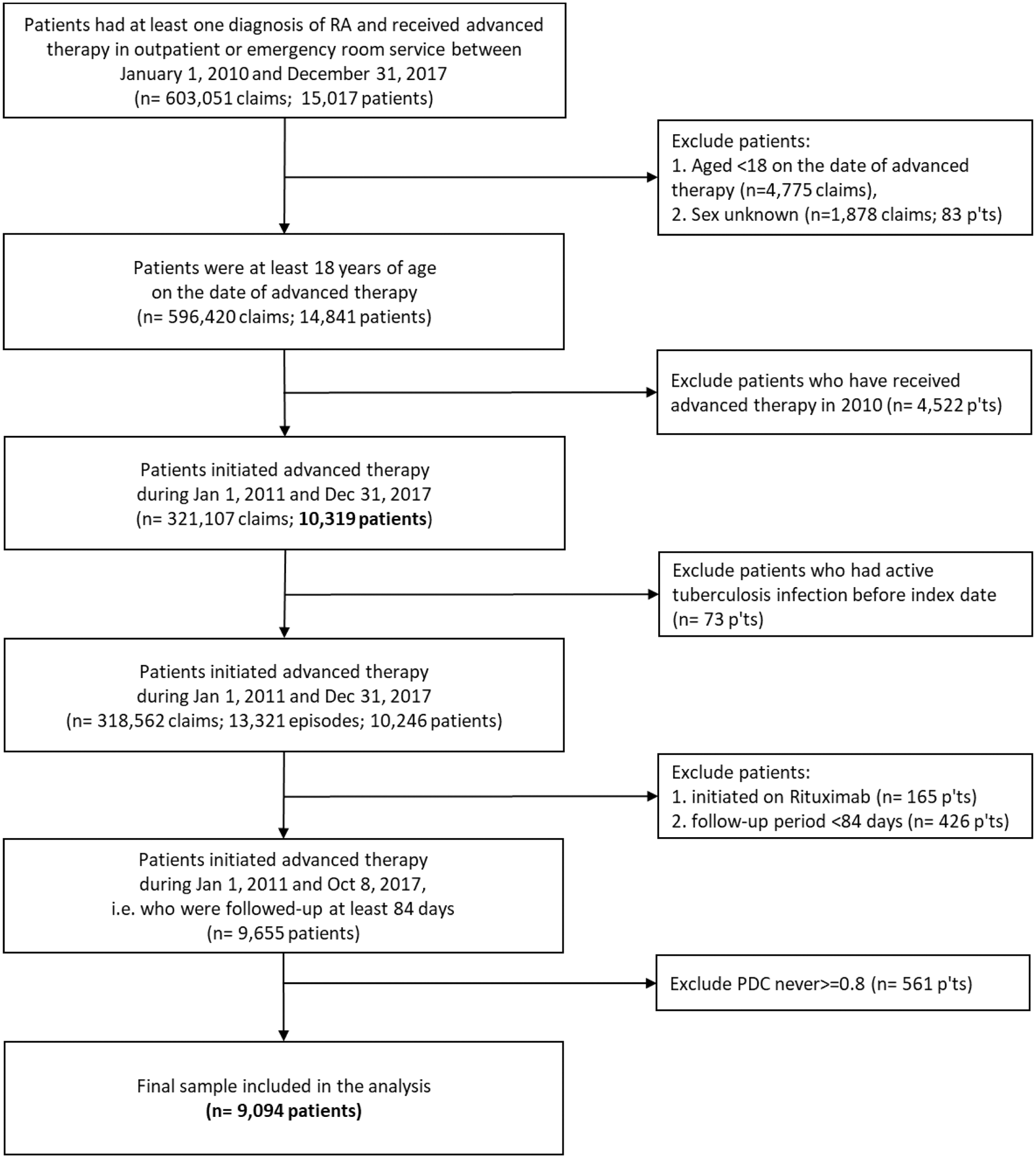
Methods: This study was an observational retrospective analysis on the population-based National Health Insurance Research Database (NHIRD) in Taiwan. Patients with RA aged ≥18, initiated an index advanced therapy - abatacept, adalimumab, etanercept, golimumab, tocilizumab, or tofacitinib, during 2011-2017 were included (
Flow chart of patient selection:
Results: The study comprised 9,094 patients initiated advance treatment for RA, with mean age of 57.3 (SD 13.3) years and 78.8% were female. The median PDC dropped remarkably after 28 months since treatment initiation (
The change of PDC of advanced therapy over the treatment period
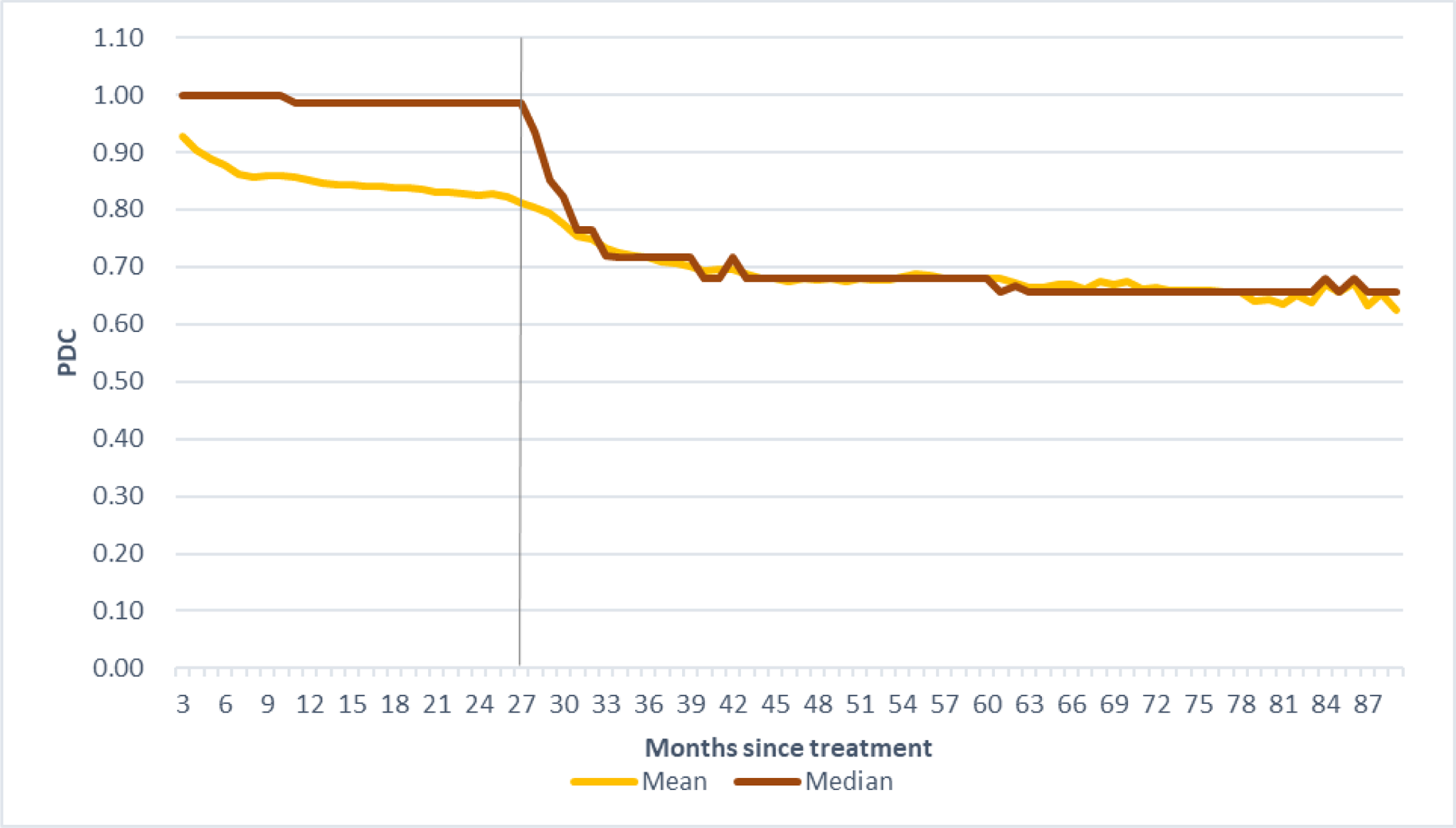
Conclusion: For RA patients, PDC of advanced therapy dropped notably after patients received advanced therapy for more than 24 months. The tapering policy implementation significantly increased the probability of dose tapering of advanced therapy in patients with treatment duration ≥ 24 months.
REFERENCES:
[1]Lenert A, Lenert P. Clin Rheumatol. 2017;36(1):1-8.
[2]Smolen JS, et al. Ann Rheum Dis. 2017;76(6):960-977.
effect of treatment duration and dose tapering policy on the probability of dose tapering
| Odds ratio | 95% | CI | p value | |
|---|---|---|---|---|
| Effect of Treatment Duration (in pre-policy period)
| 2.73 | (2.45, | 3.05) | <.001 |
| Effect of Dose Tapering policy (in treatment duration <24 months)
| 0.94 | (0.87, | 1.01) | 0.110 |
| Interaction of Treatment Duration by Dose Tapering policy
| 1.17 | (1.03, | 1.32) | 0.014 |
* Dose Tapering policy was implemented on April 1 st , 2014
Acknowledgments : Research is sponsored by Pfizer Ltd.
Disclosure of Interests: Chao-Hsiun Tang: None declared, Chia-Li Chang: None declared, Wen-Yi Shau Employee of: Pfizer, Chih-Yi Hsin Employee of: Pfizer, Ko-Jen Li Speakers bureau: Speaker fee from Pfizer, Abbvie, Roche, Bristol-Myers Squibb, Eli Lilly and Johnson & Johnson