

Background: In part 1 of the 52-week, double-blind GiACTA trial, patients with giant cell arteritis (GCA) who received weekly tocilizumab (TCZ) plus prednisone tapering reported improvement in the 36-item Short-Form Health Survey (SF-36) Mental Component Summary (MCS) and Physical Component Summary scores and FACIT-Fatigue scores that were statistically significant and clinically meaningful compared with patients who received prednisone alone. 1
Objectives: To analyze whether benefit in SF-36 MCS was maintained in patients originally assigned to TCZ compared with those originally assigned to placebo (PBO) plus a 26- or 52-week prednisone taper among patients who achieved clinical remission at week 52 and maintained treatment-free clinical remission in the 2-year, long-term extension of GiACTA.
Methods: At the end of part 1, patients entered open-label part 2, in which GCA therapy (including initiation/termination of open-label TCZ and/or GCs) was given at the investigator’s discretion according to disease status. Change from baseline in SF-36 MCS score was compared for combined original TCZ (n = 33) and PBO (n = 17) patients who achieved clinical remission at week 52 and maintained treatment-free (no TCZ or GCs) clinical remission in part 2 using a repeated-measures model. The minimal clinically important difference (MCID) for SF-36 MCS is >2.5. 2
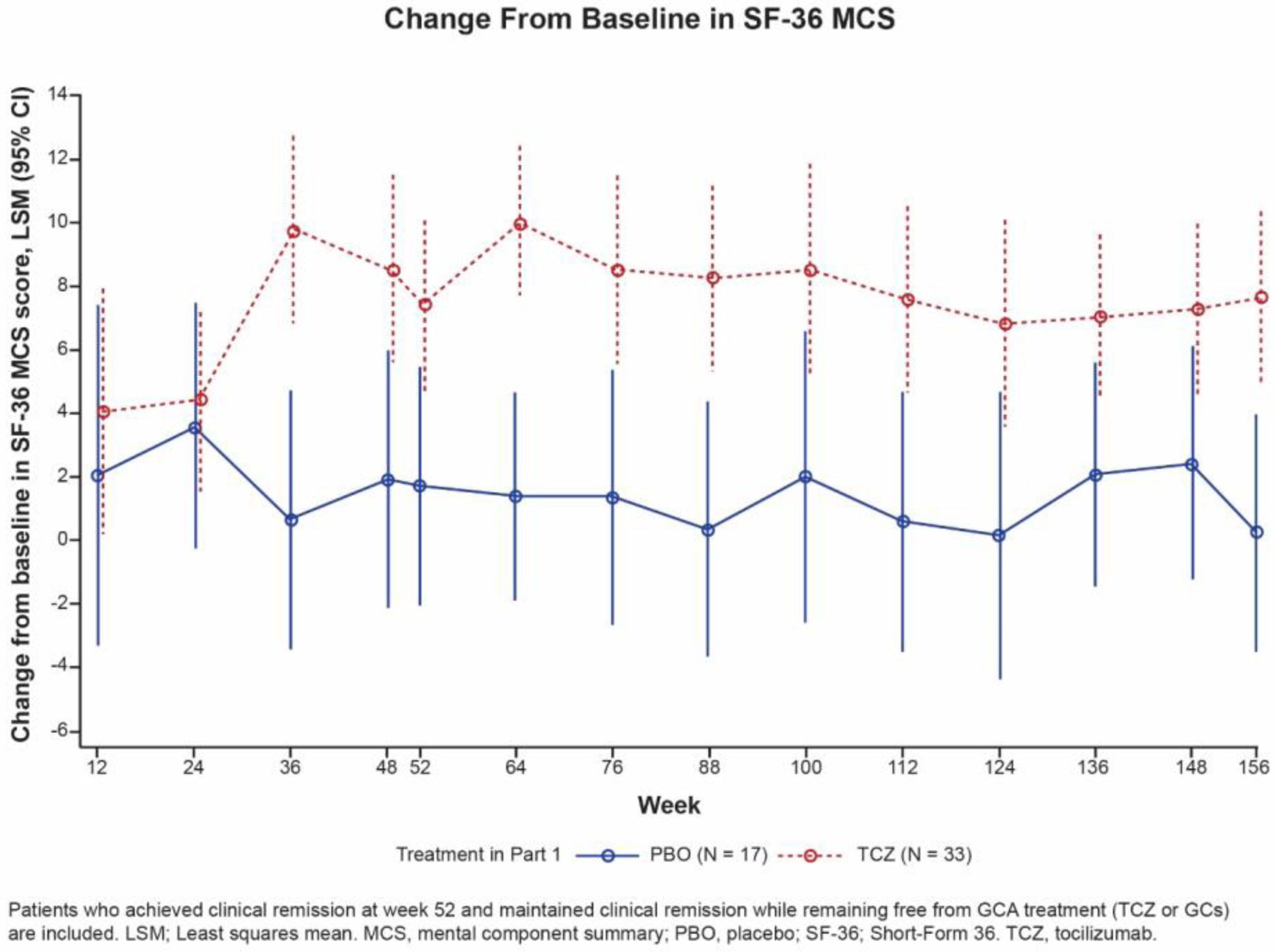
Results: During treatment, SF-36 MCS scores in all 50 patients who maintained treatment-free clinical remission in part 2 had diverged between the TCZ and PBO groups as early as 36 weeks after baseline, with greater improvements evident in the TCZ group (Figure). The difference in least square means (LSM) change between TCZ and PBO was statistically significant at week 52 ( p = 0.016) and maintained at weeks 100 ( p = 0.023) and 156 ( p = 0.002). The LSM difference (95% CI) between TCZ and PBO at weeks 52, 100, and 156 was 5.6 (1.1-10.2), 6.5 (0.9-12.1), and 7.4 (2.9-11.9), respectively, exceeding the MCID.
Conclusion: Among patients who maintained treatment-free clinical remission during part 2 of GiACTA, those originally assigned to receive TCZ plus a prednisone taper during part 1 maintained statistically significant and clinically meaningful improvements in SF-36 MCS up to week 156 compared with those originally assigned to receive PBO plus a prednisone taper in part 1. This was true even though neither of the patient groups received TCZ or GC treatment after they achieved clinical remission at week 52.
REFERENCES:
[1]Strand V et al. Arthritis Res Ther 2019;21:64.
[2]Lubeck DP. Pharmacoeconomics 2004;22:27-38.
Disclosure of Interests: John H. Stone Grant/research support from: Roche, Consultant of: Roche, Jian Han Shareholder of: Genentech, Inc., Employee of: Genentech, Inc., Sebastian Unizony Grant/research support from: Genentech, Inc., Martin Aringer Consultant of: Boehringer Ingelheim, Roche, Speakers bureau: Boehringer Ingelheim, Roche, Daniel Blockmans Consultant of: yes, Speakers bureau: yes, Elisabeth Brouwer Consultant of: Roche (consultancy fee 2017 and 2018 paid to the UMCG), Speakers bureau: Roche (2017 and 2018 paid to the UMCG), Maria C. Cid Speakers bureau: Roche, Bhaskar Dasgupta Grant/research support from: Roche, Consultant of: Roche, Sanofi, GSK, BMS, AbbVie, Speakers bureau: Roche, Jürgen Rech Consultant of: BMS, Celgene, Novartis, Roche, Chugai, Speakers bureau: AbbVie, Biogen, BMS, Celgene, MSD, Novartis, Roche, Chugai, Pfizer, Lilly, Carlo Salvarani: None declared, Robert Spiera Grant/research support from: Roche-Genetech, GSK, Boehringer Ingelheim, Chemocentryx, Corbus, Forbius, Sanofi, Inflarx, Consultant of: Roche-Genetech, GSK, CSL Behring, Sanofi, Janssen, Chemocentryx, Forbius, Mistubishi Tanabe, Min Bao Shareholder of: Roche, Employee of: Genentech