

Background: As widely demonstrated, circulating endothelial progenitor cells (EPCs) could be considered biomarkers of endothelial dysfunction. Their frequency and function varied in systemic lupus erythematosus (SLE) patients, with a significant association with subclinical atherosclerosis 1 . Caffeine, one of the most widely consumed products in the world, seems to interact with multiple components of the immune system by acting as a non-specific phosphodiesterase inhibitor 2 . In terms of cardiovascular disease (CVD), data from the literature showed a U-shaped association between habitual coffee intake and CVD 3 . In this view, Spyridopoulos et al. demonstrated a significant improvement in mature endothelial cells and EPCs migration in relation to coffee consumption in coronary artery disease both in mouse models and in patients 4 . Finally, caffeine seems to play a positive effect on SLE disease activity status, as demonstrated by the inverse association between its intake and SLE Disease Activity Index 2000 (SLEDAI-2K) and the serum levels of inflammatory cytokines 5 . At the best of our knowledge, there are no data about the effect of caffeine on cardiovascular risk in SLE patients.
Objectives: The aim of this study was to evaluate the possible role of caffeine intake on endothelial function in SLE patients, by evaluating its effect on circulating EPCs.
Methods: We performed a cross-sectional study enrolling SLE patients, fulfilling the revised 1997 ACR criteria. According with the protocol study, we excluded patients with history of smoking, CVD, chronic kidney failure, dyslipidaemia, and/or diabetes. At recruitment, the clinical and laboratory data were collected and disease activity was assessed using the SLEDAI-2k. Caffeine intake was evaluated using a 7-day food frequency questionnaire, previously employed in SLE cohort 5 . At the end of questionnaire filling, blood samples were collected. EPCs were isolated from peripheral blood mononuclear cells (PBMC) by a flow cytometry analysis and they were defined as early EPCs CD34+KDR+CD133+ cells and late EPCs CD34+KDR+CD133-, expressed as a percentage within the lymphocyte gate.
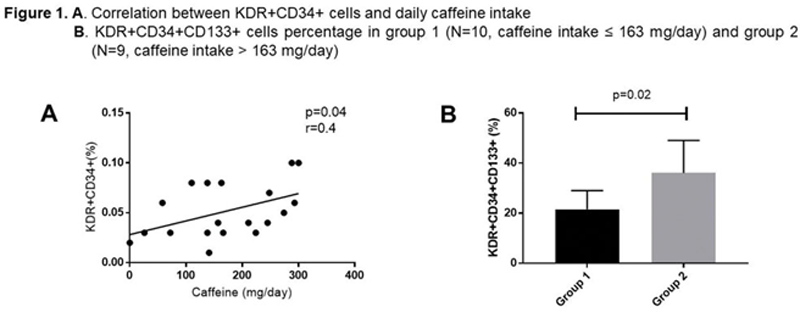
Results: We enrolled 19 patients (F:M 18:1, median age 45 years, IQR 15; median disease duration 240 months, IQR 168). In this cohort, we observed a mean±SD SLEDAI-2k value of 1.3±3.3 and the most frequent disease-related feature was joint involvement (73.7%). Concerning treatment at the time of enrolment, the majority of patients were receiving treatment with hydroxychloroquine (78.9%) and seven with glucocorticoids (36.8%). The median intake of caffeine was 163 mg/day (IQR 138) and we used this value as cut-off to categorize SLE patients in 2 groups: group 1 (N=10, caffeine intake ≤ 163 mg/day) and group 2 (N=9, caffeine intake > 163 mg/day). Patients with less intake of caffeine showed a significantly more frequent history of lupus nephritis (p=0.03), haematological manifestations (p=0.0003) and anti-dsDNA positivity (p=0.0003). Moving on EPCs, a positive correlation between caffeine intake and EPCs percentage was observed (p=0.04, r=0.4) (
Conclusion: This is the first report analysing the impact of caffeine on EPCs frequency in SLE patients. We found a positive correlation between its intake and both early and late EPCs percentage, suggesting a caffeine influence on endothelial function in SLE patients. Nonetheless, these results support the possible impact of dietary habits on autoimmune diseases.
REFERENCES:
[1]Westerweel et al. Ann Rheum Dis 2007.
[2]Aronsen et al. Europ Joul of Pharm 2014.
[3]Ding et al. Circulation 2015.
[4]Spyridopoulos et al. Art. Thromb Vasc Biol. 2008.
[5]Orefice et al. Lupus 2020.
Disclosure of Interests: None declared