

Background: Schnitzler’s syndrome is an autoinflammatory disease characterized by monoclonal gammopathy and recurrent episodes of urticaria accompanied by clinical and laboratory signs of acute inflammation. Interleukin (IL)-1 inhibitors proved to be useful in the treatment, but data on long-term safety and efficacy of these agents are sparse.
Objectives: To evaluate the retention rate of IL-1 inhibitors in patients with Schnitzler’s Syndrome.
Methods: Retrospective analysis of an Italian multicenter cohort (9 Centers). All patients fulfilled Strasbourg diagnostic criteria. Data are expressed as median [IQR].
Results: We identified 15 patients (8 females, 7 males) who received a total of 24 treatment courses with IL-1 inhibitor treatment (16 anakinra and 8 canakinumab) between January 2001 and December 2019, with a median treatment duration of 19 months [8.5-51.3]. Median age at diagnosis was 64.0 years [56.0-72.5] and median follow up was 5.0 years [2.0-8.0]. Before the biological treatment, all patients were treated with corticosteroids and 11 with at least one conventional synthetic disease-modifying antirheumatic drug (csDMARD): methotrexate (5), colchicine (5), cyclosporine (3), azathioprine (1), mycophenolate mofetil (1), cyclophosphamide (1).
Fifteen patients received 16 courses of Anakinra, which was the 1 st line biological treatment in 14 patients. Seven patients continued it with benefit, while 7 patients discontinued it: 3 for secondary inefficacy; 3 for adverse events (2 injection site reactions, 1 severe allergic reaction); 1 for secondary inefficacy and leukopenia. Anakinra was used as 2 nd line treatment in 1 case (after tocilizumab failure); in 1 patient anakinra was resumed after temporary discontinuation and an attempt with infliximab. One patient died for multiple myeloma progression while on treatment with anakinra. The median duration of the courses with anakinra was 20.0 months [6.0-58.3].
Seven patients received 8 courses of canakinumab (150 mg/8weeks in 5 cases and 150 mg/4weeks in 3). In 5 cases the drug was administered as 2 nd line biological treatment (after anakinra failure) and in 2 cases as 3 rd line treatment (1 after tocilizumab and anakinra failures and 1 after anakinra and adalimumab failure). In 1 patient, it was resumed after temporary discontinuation and an attempt with etanercept. One patient died while on treatment with canakinumab due to a presumably unrelated adverse event. The median duration of canakinumab treatment courses was 19.0 months [13.5-31.0].
At last follow-up visit, all patients were on treatment with an IL-1 inhibitor: 8 with anakinra (7 at the dosage of 100 mg/day, 1 at the dosage of 200 mg/day) and 7 with canakinumab (2 at the dosage of 150 mg/8 weeks, 4 at the dosage of 150 mg/4 weeks and 1 at the dosage of 300 mg/4 weeks). Notably, in 3 patients the dosage of canakinumab was increased since the start of the treatment.
Among 9 patients who were on treatment with prednisone at the start of the last IL-1 inhibitor, the prednisone median dose was 12.5 mg/day [10.0-18.8] while at the last follow-up visit it was 5.0 mg/day [0-7.5] (p= 0.02).
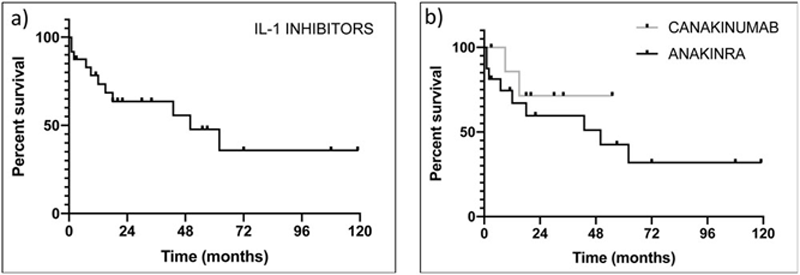
The retention rate of IL-1 inhibitors was 73.4% [SE 9.4] at 1 year and 63.6% [SE 10.4] at 2 years (
a) Retention rate of IL-1 inhibitors (24 courses); b) Retention rate of canakinumab (8 courses) and anakinra (16 courses).
Conclusion: In this multicentric cohort of patients affected by Schnitzler’s syndrome, the treatment with IL-1 inhibitors as 1 st , 2 nd or 3 rd line biological treatment permitted a good disease control and corticosteroid reduction in patients who did not respond to csDMARDs and/or to prior other biological DMARDs. The optimal dosage of these drugs needs to be tailored for every patient.
Acknowledgements: AIDA Network
Disclosure of Interests: Francesca Crisafulli: None declared, Antonio Vitale: None declared, Carla Gaggiano: None declared, Lorenzo Dagna: None declared, Giulio Cavalli Speakers bureau: SOBI, Novartis, Paid instructor for: SOBI, Novartis, Consultant of: SOBI, Novartis, Rolando Cimaz: None declared, Ombretta Viapiana: None declared, Florenzo Iannone: None declared, Giuseppe Lopalco: None declared, Roberto Bortolotti: None declared, Masen Abdel Jaber: None declared, Carlomaurizio Montecucco: None declared, Sara Monti: None declared, Silvia Balduzzi: None declared, Giacomo Emmi: None declared, Paolo Airò: None declared, Franco Franceschini: None declared, Luca Cantarini Speakers bureau: SOBI, Novartis, Paid instructor for: SOBI, Grant/research support from: SOBI, Novartis, Micol Frassi: None declared