

Background: Baricitinib (BARI), an oral, selective Janus kinase (JAK)1 and JAK2 inhibitor, improved disease severity in adults with systemic lupus erythematosus (SLE) receiving standard background therapy in a phase 2 trial 1 . There were no meaningful reductions in least squares mean change from baseline (BL) in levels of serologic biomarkers for SLE with BARI treatment, including anti-double-stranded deoxyribonucleic acid (anti-dsDNA) antibodies and complement component (C)3 and C4 1 .
Objectives: Evaluate the median change from BL in serologic biomarkers in subgroups and the overall population of BARI-treated SLE patients, in addition to the Systemic Lupus Erythematosus Responder Index-4 (SRI-4) response by normalization of anti-dsDNA.
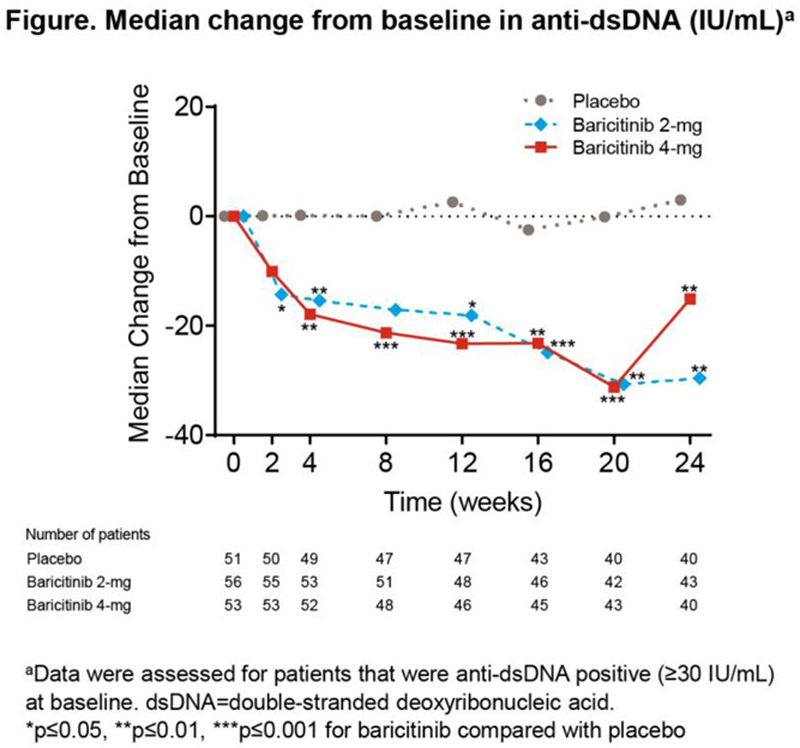
Methods: Data were assessed from the phase 2 trial JAHH (NCT02708095). The median change from BL in anti-dsDNA, IgG, C3, and C4 was evaluated over time among the following populations at BL: anti-dsDNA positive (≥30 IU/mL), low C3 (<90 mg/dL), low C4 (<10 mg/dL), and all patients for IgG. Statistical tests were conducted for BARI 2-mg and 4-mg compared with placebo (PBO). Among patients who were anti-dsDNA positive at BL, SRI-4 responder rate was compared for those who stayed positive or achieved normal levels by Week (Wk) 24.
Results: Among patients who were anti-dsDNA positive at BL, significant decreases of anti-dsDNA antibodies were observed for BARI 2-mg and 4-mg compared to PBO beginning at Wks 2 and 4, respectively, and continuing through Wk 24 (
Conclusion: BARI treatment resulted in a rapid and sustained significant decrease in anti-dsDNA antibodies compared to PBO among anti-dsDNA positive SLE patients at BL, as well as a significant decrease in IgG levels in the 4-mg group at Wks 12 and 24. These data suggest that BARI may have an effect on B cell activity in SLE.
REFERENCES:
[1]Wallace D et al. Lancet. 2018;392:222-231.
| PBO | BARI 2-mg | BARI 4-mg | |||||||
| Week | 4 | 12 | 24 | 4 | 12 | 24 | 4 | 12 | 24 |
| Anti-dsDNA (IU/mL ) a | 0.2 (-17.2, 17.3) | 2.6 (-14.8, 18.4) | 3.0 (-14.9, 28.3) | -15.4** (-31.4, 1.9) | -18.1* (-42.0, 4.1) | -29.6** (-55.1, 10.3) | -17.9** (-42.7, 1.8) | -23.3*** (-50.9, -5.9) | -15.1** (-71.9, -4.6) |
| IgG (g/L ) b | -0.31 (-1.1, 0.4) | 0.09 (-1.1, 0.7) | -0.04 (-0.9, 0.9) | -0.60 (-1.3, 0.6) | -0.30 (-1.3, 0.4) | -0.51 (-1.7, 0.6) | -0.56 (-1.2, 0.2) | -0.65** (-1.3, 0.2) | -0.60** (-1.7, 0.2) |
Data are median change from baseline (25 th , 75 th percentiles). a Data were assessed for patients that were anti-dsDNA positive (≥30 IU/mL) at baseline (PBO N=51, BARI 2-mg N=56, BARI 4-mg N=53). b Data were assessed for all patients (PBO N=105, BARI 2-mg N=105, BARI 4-mg N=104). *p≤0.05, **p≤0.01, ***p≤0.001 for BARI vs. PBO.
Acknowledgements: The authors would like to acknowledge Nicole L. Byers, of Eli Lilly and Company, for medical writing and process support.
Disclosure of Interests: Thomas Dörner Speakers bureau: Eli Lilly and Company, Roche, and Samsung, Consultant of: AbbVie, Celgene, Eli Lilly and Company, Janssen, Novartis, Roche, Samsung, and UCB, Grant/research support from: Janssen, Novartis, Roche, Sanofi, and UCB, Ronald van Vollenhoven Consultant of: Abbvie, Biotest, BMS, Celgene, Crescendo, Eli Lilly and Company, GSK, Janssen, Merck, Novartis, Pfizer, Roche, UCB, and Vertex, Grant/research support from: Abbvie, Amgen, BMS, GSK, Pfizer, Roche, and UCB, Andrea Doria Speakers bureau: GSK, Janssen, Pfizer, and Roche, Consultant of: Celgene, Eli Lilly and Company, and GSK, Bochao Jia Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Damiano Fantini Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Jorge Ross Terres Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Maria Silk Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Stephanie de Bono Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Peter Fischer Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Daniel J. Wallace Consultant of: Amgen, Aurunia, Eli Lilly and Company, EMD Merck Serono, GSK, and Pfizer