

Background: In the TULIP-1 and TULIP-2 trials, anifrolumab, a type I interferon (IFN) receptor antibody, at a dosage of 300 mg once every 4 weeks (Q4W), demonstrated consistent median pharmacokinetic (PK) concentrations 1 and sustained neutralization of the pharmacodynamic (PD) 21-gene type I IFN gene signature (IFNGS) 2–4 in patients with moderate to severe systemic lupus erythematosus (SLE) despite standard therapy.
Objectives: To characterize the PK/PD relationship of anifrolumab and to confirm anifrolumab 300 mg provides adequate PD neutralization in IFNGS test–high patients.
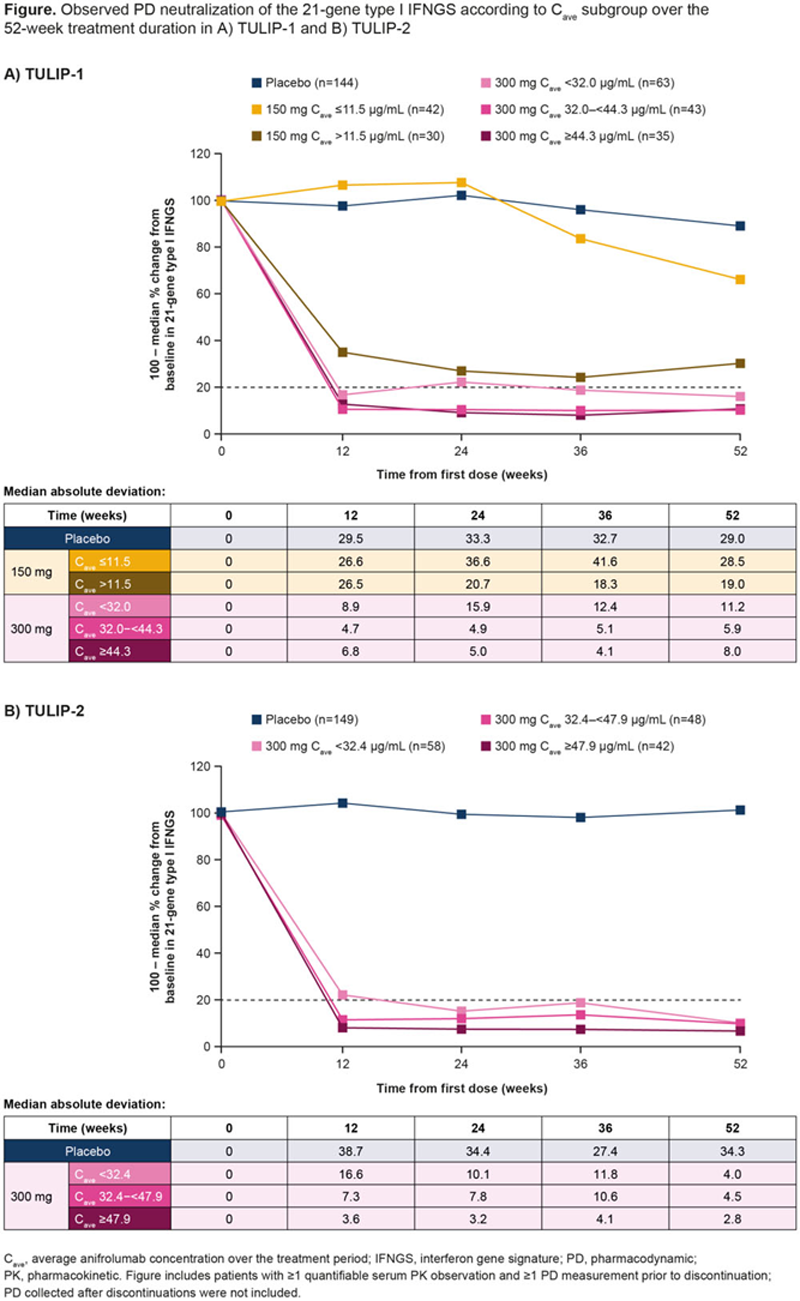
Methods: This study included IFNGS test–high patients from the phase 3 randomized, placebo-controlled, 52-week TULIP-1 2 (NCT02446912) and TULIP-2 3 (NCT02446899) trials of intravenous anifrolumab 150 mg or 300 mg Q4W plus standard therapy. IFNGS test status (high or low) at screening was classified with an analytically validated 4-gene qPCR based test on whole blood. 2 PD neutralization was measured with 21-gene type I IFNGS and expressed as a percentage change from baseline. 3 For the graphic PK/PD analysis, patients with ≥1 quantifiable serum PK sample and ≥1 PD measurement before discontinuation were categorized depending on C ave (individual predicted average anifrolumab concentration over treatment duration) median and tertiles (T) for anifrolumab 150 mg and 300 mg, respectively. Median PD IFNGS neutralization and medium absolute deviations were compared across C ave subgroups. PK/PD modeling was assessed in patients with ≥1 quantifiable serum PK sample and a baseline and ≥1 postbaseline PD measurement before discontinuation, using a nonlinear mixed-effects model (NONMEM; version 7.3; ICON) to estimate parameters and characterize the PK/PD data. The PD/efficacy analysis included patients with ≥1 postbaseline PD measurement before discontinuation. BILAG-based Combined Lupus Assessment (BICLA) response rates at Week (W)52 were compared across median PD neutralization quartiles (Q) for pooled anifrolumab 300 mg and 150 mg groups.
Results: The PK/PD graphic analysis included 654 IFNGS test–high patients (placebo [n=293]; anifrolumab 150 mg [n=72] or 300 mg [n=289]). C
ave
was generally higher with anifrolumab 300 mg (µg/mL, TULIP-1: T1 <32, T2 32–<44.3, T3 ≥44.3; TULIP-2: T1 <32.4, T2 32.4–<47.9, T3 ≥47.9) than with anifrolumab 150 mg (median 11.5 µg/mL); overlap between anifrolumab 300 mg and 150 mg C
ave
subgroups was small owing to nonlinearity. Anifrolumab 300 mg elicited rapid (by W4
4
) and sustained median PD neutralization >80%, vs a lower and delayed PD neutralization (median >50% at W52) with anifrolumab 150 mg, and minimal PD neutralization with placebo. The median PD neutralization increased with higher C
ave
subgroups, plateauing at ~90% at W12–W52. All anifrolumab 300 mg C
ave
tertiles had a median PD neutralization ~80%; however, the variability was greater in the lowest C
ave
tertiles vs higher C
ave
tertiles across trials (
Conclusion: In TULIP-1 and TULIP-2, anifrolumab 300 mg yielded higher anifrolumab C ave vs 150 mg. High C ave was associated with rapid (W4 4 –W12), substantial, and sustained PD neutralization of the 21-gene IFNGS in IFNGS test–high patients, which in turn was associated with higher efficacy.
REFERENCES:
[1]Kuruvilla D. Poster 360, AAPS 2020.
[2]Furie RA. Lancet Rheumatol. 2019;1:e208–19.
[3]Morand EF. N Engl J Med. 2020;382:211–21.
[4]Furie R. Arthritis Rheumatol. 2017;69:379–86.
Acknowledgements: Writing assistance by Matilda Shackley, MPhil, of JK Associates Inc., part of Fishawack Health. This study was sponsored by AstraZeneca.
Disclosure of Interests: Yen Lin Chia Employee of: AstraZeneca, Raj Tummala Employee of: AstraZeneca, Tu Mai Employee of: Genentech, Tomas Rouse Employee of: AstraZeneca, Wendy White Employee of: AstraZeneca, Eric F. Morand Speakers bureau: AstraZeneca, Consultant of: AstraZeneca, Grant/research support from: AstraZeneca, Richard Furie Consultant of: AstraZeneca, Grant/research support from: AstraZeneca