

Background: Anifrolumab, a type I interferon receptor antibody, has shown efficacy in patients with systemic lupus erythematosus (SLE), 1,2 >30% of whom develop lupus nephritis (LN).
Objectives: To evaluate the efficacy and safety of anifrolumab vs placebo alongside standard therapy in patients with active proliferative LN.
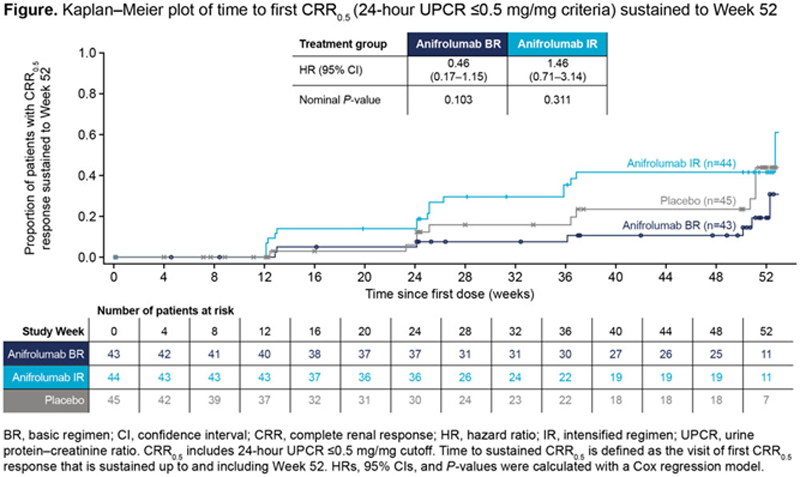
Methods: TULIP-LN (NCT02547922) was a phase 2 double-blind trial in adult patients with active, biopsy-proven LN and 24-hour (h) urine protein–creatinine ratio (UPCR) >1 mg/mg. Patients were randomized (1:1:1) to anifrolumab basic regimen (BR, 300 mg, based on SLE dosing 1,2 ), anifrolumab intensified regimen (IR, 900 mg for 3 doses, 300 mg thereafter), or placebo, intravenously every 4 weeks alongside standard therapy of oral glucocorticoids (GCs; mandatory taper ≤10 mg/day by Week [W]12, ≤7.5 mg/day by W24) and mycophenolate mofetil (target 2 g/day by W8). The primary endpoint was the relative difference in change from baseline to W52 in 24-h UPCR, measured with a geometric mean ratio (GMR) of the change in the combined anifrolumab vs placebo groups (GMR <1 favors anifrolumab). The key secondary endpoint was complete renal response (CRR) at W52 (24-h UPCR ≤0.7 mg/mg, estimated glomerular filtration rate ≥60 mL/min/1.73 m 2 or no decrease ≥20%, no treatment discontinuation, and no restricted medication use). Sustained GC taper (≤7.5 mg/day, W24–52) was an exploratory endpoint. CRR 0.5 (CRR with UPCR ≤0.5 mg/mg) and time to CRR 0.5 sustained to W52 were analyzed post hoc. Responder rates were calculated with a stratified Cochran–Mantel–Haenszel approach.
Results: Patients received anifrolumab BR (n=45) or IR (n=51) or placebo (n=49); demographics and baseline disease characteristics were generally balanced between groups. No difference in change from baseline to W52 in 24-h UPCR was observed for combined anifrolumab vs placebo groups (
Conclusion: Although the primary endpoint was not met, the anifrolumab IR was associated with numeric improvements across clinical endpoints vs placebo; thus, intensified dosing may be required to reach clinical efficacy in LN vs SLE without active renal disease. Anifrolumab had a similar safety profile in patients with LN and SLE; despite higher frequency of HZ vs placebo, anifrolumab was well tolerated.
REFERENCES:
[1]Morand EF. N Engl J Med . 2020;382:211–21.
[2]Furie RA. Lancet Rheumatol. 2019;1:e208–19.
Summary of Clinical Efficacy Endpoints
| Endpoint | Anifrolumab | Placebo | |||
| Combined | Basic | Intensified | |||
| 24-hour urine protein–creatinine ratio improvement W52 | N | 91 | 41 | 50 | 41 |
|
GMR vs placebo
| 1.031 | 1.104 | 0.963 | – | |
| 95% CI | 0.621, 1.713 | 0.612, 1.992 | 0.548, 1.693 | ||
| CRR rate W52 | n/N (% ) | 27/87 (31.0) | 7/43 (16.3) | 20/44 (45.5) | 14/45 (31.1) |
| Δ | −0.08 | −14.83 | 14.34 | – | |
| 95% CI | −16.92, 16.76 | −32.89, 3.22 | −5.77, 34.46 | ||
| CRR 0.5 rate W52 | n/N (% ) | 25/87 (28.7) | 7/43 (16.3) | 18/44 (40.9) | 12/45 (26.7) |
| Δ | 2.07 | −10.39 | 14.24 | – | |
| 95% CI | −14.25, 18.39 | −28.07, 7.29 | −5.42, 33.90 | ||
|
Glucocorticoid
| n/N (% ) | 31/67 (46.3) | 11/31 (35.5) | 20/36 (55.6) | 11/33 (33.3) |
| Δ | 12.94 | 2.15 | 22.22 | – | |
| 95% CI | −7.26, 33.13 | −21.40, 25.70 | −0.79, 45.23 | ||
Δ Percentage difference vs placebo.
CI, confidence interval; CRR, complete renal response; GMR, geometric mean ratio; n, number of responders; N, number analyzed; W, Week.
Acknowledgements: Writing assistance by Matilda Shackley, MPhil, of JK Associates, Inc, a member of Fishawack Health. This study was sponsored by AstraZeneca.
Disclosure of Interests: David Jayne Grant/research support from: AstraZeneca, Aurinia, Boehringer-Ingelheim, GSK, Roche/Genentech and Sanofi-Genzyme, Brad H Rovin Consultant of: AstraZeneca, Eduardo Mysler Grant/research support from: AstraZeneca, GSK, Eli Lilly, Sandoz, Roche, AbbVie, Pfizer, Janssen, Gemma, and Amgen, Richard Furie Consultant of: AstraZeneca, Grant/research support from: AstraZeneca, Frederic Houssiau Consultant of: GSK, Teodora Trasieva Employee of: AstraZeneca, Jacob Knagenhjelm Employee of: AstraZeneca, Erik Schwetje Employee of: AstraZeneca, Yen Lin Chia Employee of: AstraZeneca, Raj Tummala Employee of: AstraZeneca, Catharina Lindholm Employee of: AstraZeneca