

Background: Type I interferons such as Anifrolumab have been implicated in Systemic lupus erythematosus (SLE) pathogenesis on the basis of increased interferon-stimulated gene expression and genetic susceptibility. Little is known regarding its efficacy and safety profile.
Objectives: To assess the efficacy and safety of Anifrolumab in patients with SLE.
Methods: Electronic databases (PubMed, Embase, Scopus, Cochrane) were searched from inception until December 15th, 2020. Unadjusted odds ratios (OR) were calculated from dichotomous data using Mantel Haenszel (M-H) random-effects with statistical significance to be considered if the confidence interval excludes 1 and p<0.05. The primary outcome of interest was British Isles Lupus Assessment Group (BILAG)-based Composite Lupus Assessment (BICLA). Secondary outcomes included the proportion of patients who achieved an SLE responder index of 4 (SRI-4) reduction of 50% or more in the Cutaneous Lupus Erythematosus Disease Area and Severity Index (CLASI), reductions in the glucocorticoid dose and adverse effects.
Results: A total of three studies 1,2,3 with 839 participants (Anifrolumab=372, Placebo=467) were included in our analysis. Follow-up duration was at week 52. A statistically significant different was observed in the Anifrolumab arm in terms of BICLA response (OR 0.44 95%CI 0.34-0.59;p < 0.00001, I 2 =4), ≥50% reduction in CLASI activity score (OR 0.36 95%CI 0.21-0.60;p=0.0001, I 2 =0), glucocorticoid reduction (OR 0.41 95%CI 0.28-0.59;p<0.00001; I 2 =0) and SRI-4 response (OR 0.52 95% CI 0.30-0.90; p=0.02, I 2 =75). However, Adverse events were less likely in the placebo arm as compared to Anifrolumab (OR 1.54 95%CI 1.05-2.25; p=0.03; I 2 =0).
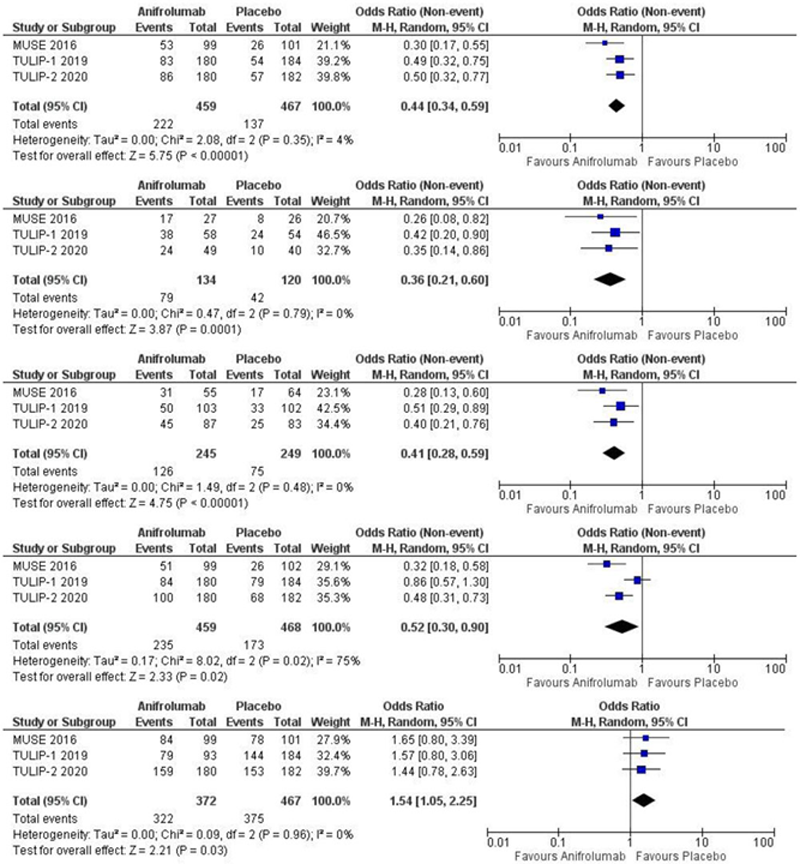
Conclusion: Anifrolumab was found to be more effective than placebo for the management of SLE, but may also cause more severe adverse effects.
REFERENCES:
[1]Morand EF, Furie R, Tanaka Y, Bruce IN, Askanase AD, Richez C, Bae SC, Brohawn PZ, Pineda L, Berglind A, Tummala R; TULIP-2 Trial Investigators. Trial of Anifrolumab in Active Systemic Lupus Erythematosus. N Engl J Med. 2020 Jan 16;382(3):211-221. doi: 10.1056/NEJMoa1912196. Epub 2019 Dec 18. PMID: 31851795.
[2]Furie R, Khamashta M, Merrill JT, Werth VP, Kalunian K, Brohawn P, Illei GG, Drappa J, Wang L, Yoo S; CD1013 Study Investigators. Anifrolumab, an Anti-Interferon-α Receptor Monoclonal Antibody, in Moderate-to-Severe Systemic Lupus Erythematosus. Arthritis Rheumatol. 2017 Feb;69(2):376-386. doi: 10.1002/art.39962. PMID: 28130918; PMCID: PMC5299497.
[3]Furie RA, Morand EF, Bruce IN, et al. Type I interferon inhibitor anifrolumab in active systemic lupus erythematosus (TULIP-1): a randomised, controlled, phase 3 trial. Lancet Rheumatol 2019; 1(4):e208-e219.
Disclosure of Interests: None declared