

Background: Tocilizumab (TCZ) showed trends for improving skin fibrosis and prevented progression of lung fibrosis in patients with systemic sclerosis (SSc) in placebo-controlled randomised clinical trials (RCTs). However, safety and effectiveness of TCZ beyond these selected and enriched clinical trial populations in SSc is still unknown.
Objectives: To assess safety and effectiveness of TCZ treatment compared to standard of care in SSc patients from the large, multicentre, observational, real-life EUSTAR network/database using propensity score matching.
Methods: SSc patients from the EUSTAR network/database, who fulfilled the ACR/EULAR 2013 classification criteria, with a baseline and a follow-up visit at 12±3 months, receiving TCZ or standard of care (controls), were selected. The following variables were used for the propensity score matching (1:1): age at diagnosis, gender, disease subtype, baseline modified Rodnan skin score (mRSS), forced vital capacity (FVC), and diffusing capacity for carbon monoxide (DLCO), co-therapy with immunosuppressives, disease duration, and year of treatment. Primary endpoints were mRSS and FVC at 12±3 months follow-up compared between the groups, using paired t-tests. Secondary endpoints were the percentage of progressive/regressive patients for skin and lung at 12±3 months follow-up according to standard definitions (1,2). Sensitivity analyses assessed pre-processing decisions (selection of most recent vs. random observation for control patients with multiple suitable time intervals), as well as the matching method (optimal vs. exact matching). Missing values were addressed with 100-fold multiple imputation using chained equations. Safety data were analysed in all patients. The study including the statistical analysis plan was pre-registered at
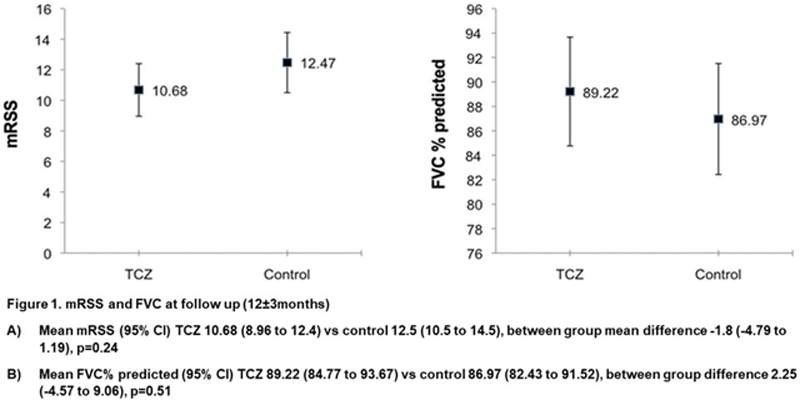
Results: We identified 93 SSc patients treated with TCZ and 2370 SSc patients with standard of care who fulfilled the inclusion criteria. Forty nine (57.7%) of the TCZ treated patients were diffuse, eight patients were not classified, disease duration was (mean±SD) 6.35±5.40 years, their baseline mRSS was 15.05±10.85, and 76 (81.7%) received immunosuppressive therapy in addition to TCZ.Through multiple imputation and propensity score matching, 100 imputed sets of 93 pairs of TCZ/controls were generated. Comparison between groups showed consistent effects of TCZ across all pre-defined primary and secondary endpoints: mRSS was lower in the TCZ group (mean difference (95% confidence interval (CI)) -1.8 (-4.79 to 1.19), p=0.24,
Conclusion: In this large, observational, controlled, real-life EUSTAR study, effectiveness of TCZ did not reach statistical significance compared to standard of care treatment but showed consistent positive effects of TCZ on skin and lung fibrosis across all pre-defined primary and secondary endpoints confirming data from recent RCTs.
REFERENCES:
[1]Prediction of improvement in skin fibrosis in diffuse cutaneous systemic sclerosis: a EUSTAR analysis. Ann Rheum Dis 2016:1743-8.
[2]Progressive interstitial lung disease in patients with systemic sclerosis-associated interstitial lung disease in the EUSTAR database. Ann Rheum Dis 2021:219-227.
Disclosure of Interests: Simon Kuster: None declared, Suzana Jordan: None declared, Muriel Daniele Elhai: None declared, Ulrike Held: None declared, Klaus Steigmiller: None declared, Cosimo Bruni: None declared, Florenzo Iannone: None declared, Serena Vettori: None declared, Elise Siegert: None declared, Simona Rednic: None declared, Veronica Codullo: None declared, Paolo Airò Consultant of: Dr. Airo’ reports personal fees (consultancies) from Bristol Myers Squibb, Bohringer Ingelheim, non-financial support from CSL Behring, SOBI, Janssen, Roche, Sanofi, Pfizer, Yolanda Braun-Moscovici: None declared, Nicolas Hunzelmann: None declared, Maria Joao Salvador: None declared, Valeria Riccieri: None declared, Ana Maria Gheorghiu: None declared, Juan Jose Alegre Sancho: None declared, Katarzyna Romanowska-Prochnicka: None declared, Ivan Castellví: None declared, Ina Koetter: None declared, Marie-Elise Truchetet Consultant of: Marie-Elise Truchetet has had consultancy relationships and/or has received research funding from Boehringer Ingelheim, Genentech/Roche, and Sanofi in the area of potential treatments of scleroderma and its complications., Grant/research support from: Marie-Elise Truchetet has had consultancy relationships and/or has received research funding from Boehringer Ingelheim, Genentech/Roche, and Sanofi in the area of potential treatments of scleroderma and its complications., Francisco J López-Longo: None declared, Pavel Novikov: None declared, Alessandro Giollo: None declared, Yuichiro Shirai: None declared, Laura Belloli: None declared, Elisabetta Zanatta: None declared, Eric Hachulla: None declared, Vanessa Smith: None declared, Christopher Denton: None declared, Ruxandra Ionescu: None declared, Tim Schmeiser: None declared, Jörg H.W. Distler: None declared, Armando Gabrielli: None declared, Anna-Maria Hoffmann-Vold Consultant of: AMHV has received research funding and/or consulting fees and/or other remuneration from Actelion, Boehringer Ingelheim, Roche, Bayer, Merck Sharp & Dohme, ARXX, Lilly and Medscape, Grant/research support from: AMHV has received research funding and/or consulting fees and/or other remuneration from Actelion, Boehringer Ingelheim, Roche, Bayer, Merck Sharp & Dohme, ARXX, Lilly and Medscape. Masataka Kuwana: None declared, Yannick Allanore: None declared, Oliver Distler Speakers bureau: Oliver Distler has/had consultancy relationship and/or has received research funding in the area of potential treatments for systemic sclerosis and its complications from (last three years): Abbvie, Acceleron Pharma, Amgen, AnaMar, Arxx Therapeutics, Baecon Discovery, Blade Therapeutics, Bayer, Boehringer Ingelheim, ChemomAb, Corbus Pharmaceuticals, CSL Behring, Galapagos NV, Glenmark Pharmaceuticals, GSK, Horizon (Curzion) Pharmaceuticals, Inventiva, iQvia, Italfarmaco, iQone, Kymera Therapeutics, Lilly, Medac, Medscape, Mitsubishi Tanabe Pharma, MSD, Novartis, Pfizer, Roche, Sanofi, Serodapharm, Topadur, Target Bioscience and UCB. Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143)., Consultant of: Oliver Distler has/had consultancy relationship and/or has received research funding in the area of potential treatments for systemic sclerosis and its complications from (last three years): Abbvie, Acceleron Pharma, Amgen, AnaMar, Arxx Therapeutics, Baecon Discovery, Blade Therapeutics, Bayer, Boehringer Ingelheim, ChemomAb, Corbus Pharmaceuticals, CSL Behring, Galapagos NV, Glenmark Pharmaceuticals, GSK, Horizon (Curzion) Pharmaceuticals, Inventiva, iQvia, Italfarmaco, iQone, Kymera Therapeutics, Lilly, Medac, Medscape, Mitsubishi Tanabe Pharma, MSD, Novartis, Pfizer, Roche, Sanofi, Serodapharm, Topadur, Target Bioscience and UCB. Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143)., Grant/research support from: The study was partially supported by a grant from Roche. Roche was not involved in analysis or interpretation of the results.