

Background: In the DISCOVER-1 study, the interleukin-23 p19 subunit inhibitor guselkumab (GUS) demonstrated robust efficacy across joint and skin clinical manifestations of psoriatic arthritis (PsA). 1 Patients (pts) with PsA also experience a broad range of symptoms that negatively impact health-related quality of life (eg, pain, fatigue, anxiety, depression, sleep disturbance, poor physical function). 2
Objectives: Assess the treatment effect of GUS on general health outcomes in pts with PsA in the DISCOVER-1 trial through Week (W) 52 using the Patient-Reported Outcomes Measurement Information System-29 (PROMIS-29) instrument.
Methods: Pts with active PsA (≥3 swollen + ≥3 tender joints; C-reactive protein ≥0.3 mg/dL) and inadequate response to standard conventional therapies were randomized 1:1:1 to GUS 100 mg Q4W; GUS 100 mg at W0, W4, then Q8W; or placebo (PBO). PBO pts switched to GUS 100 mg Q4W at W24. PROMIS-29 contains 4 items for each of 7 domains (anxiety, depression, fatigue, pain interference, physical function, sleep disturbance, social participation) and 1 pain intensity item; 28 items are scored on a 5-point Likert-type scale, and pain intensity is rated from 0-10. The raw score of each domain is converted to a standardized T-score, with norms based on a general population mean score=50 and a standard deviation (SD)=10. Higher scores in anxiety, depression, fatigue, pain interference, and sleep disturbance indicate more severe symptoms; higher physical function and social participation scores indicate better health outcomes. Changes ≥5 points (1/2 SD of T-score) are considered clinically meaningful. Analyses were performed using both observed (mean scores/changes, effect sizes) and imputed (clinically meaningful response, whereby change from baseline was set to 0 at W24/52 for pts who had missing data or at W24 for pts who met treatment failure criteria prior to W24).
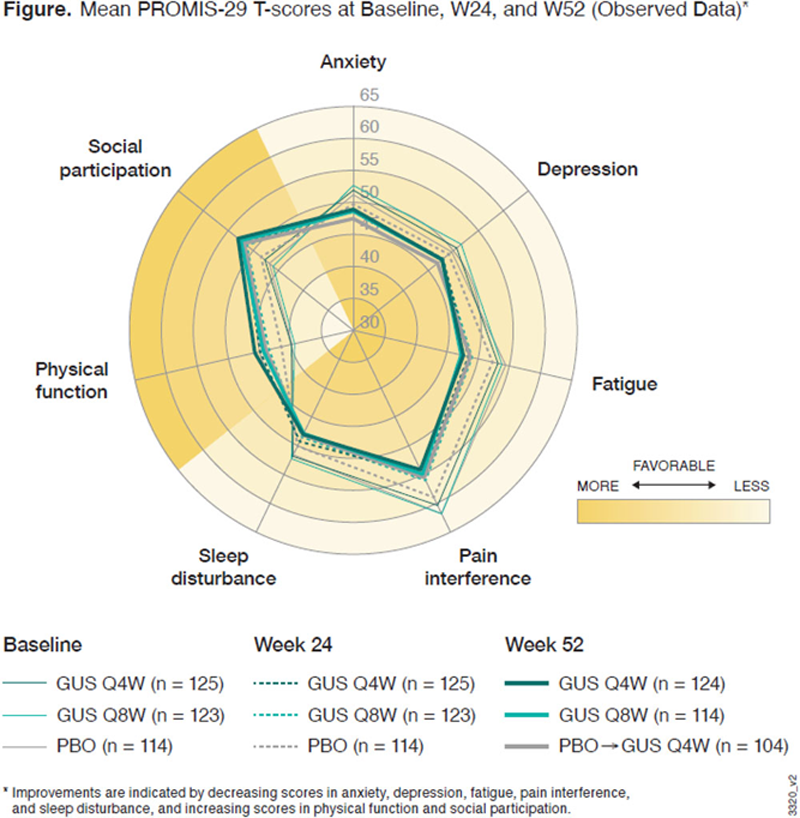
Results: At baseline, mean PROMIS-29 T-scores for physical function, social participation, sleep disturbance, pain, and fatigue were worse in the 381 PsA pts enrolled in DISCOVER-1 than in the general US population. Across all 7 domains, observed mean PROMIS-29 T-scores showed improvements in GUS-treated pts from baseline to W24 and W52 (
Conclusion: In pts with active PsA, PROMIS-29 results indicate that GUS treatment was associated with clinically meaningful reductions in fatigue and pain and improvement in physical function and social participation, which were maintained through 1 year.
REFERENCES:
[1] Deodhar A et al. Lancet. 2020;395:1115-25.
[2] Orbai A et al. Ann Rheum Dis. 2017;76:673-80.
Mean Change and Effect Size of Change From Baseline inPROMIS-29 Domain Scores at W24 and W52 (Observed)
| Mean Change From Baseline [Effect Size] | ||||||
| GUS Q4W | GUS Q8W |
PBO
|
GUS Q4W
|
|||
| W24 | W52 | W24 | W52 | W24 | W52 | |
| Anxiety | −3.1 [−0.3] | −3.1 [−0.3] | −3.7 [−0.4] | −4.3 [−0.5] | −1.5 [−0.2] | −3.6 [−0.4] |
| Depression | −2.7 [−0.3] | −3.0 [−0.4] | −4.0 [−0.4] | −4.0 [−0.4] | −0.6 [−0.1] | −2.5 [−0.3] |
| Fatigue | −4.8 [−0.5] | −5.6 [−0.6] | −4.8 [−0.5] | −6.8 [−0.7] | −2.1 [−0.2] | −5.7 [−0.6] |
| Pain interference | −5.4 [−0.8] | −6.2 [−1.0] | −5.8 [−1.0] | −7.0 [−1.1] | −2.8 [−0.4] | −6.3 [−1.0] |
| Physical function | 5.0 [0.8] | 5.9 [0.9] | 4.1 [0.6] | 5.0 [0.7] | 1.7 [0.2] | 4.2 [0.6] |
| Sleep disturbance | −2.5 [−0.4] | −3.9 [−0.6] | −3.8 [−0.6] | −4.4 [−0.6] | −1.5 [−0.2] | −3.3 [−0.5] |
| Social participation | 4.2 [0.5] | 5.3 [0.7] | 5.3 [0.6] | 6.6 [0.8] | 1.7 [0.2] | 4.9 [0.6] |
| Pain intensity* | −2.3 [−1.2] | −2.8 [−1.5] | −2.1 [−1.1] | −2.7 [−1.4] | −0.7 [−0.4] | −2.5 [−1.3] |
*Raw score; all other domains reported as T-score.
Disclosure of Interests: Ana-Maria Orbai Consultant of: Eli Lilly, Janssen, Novartis, Pfizer, UCB, Grant/research support from: Abbvie, Eli Lilly and Company, Celgene, Novartis, Janssen, Horizon, Laura C Coates Consultant of: AbbVie, Amgen, Biogen, Bristol Myers Squibb, Boehringer Ingelehim, Celgene, Domain, Eli Lilly, Gilead, Janssen, Medac, Novartis, Pfizer and UCB, Grant/research support from: AbbVie, Amgen, Celgene, Eli Lilly, Gilead, Novartis, Pfizer, Atul Deodhar Speakers bureau: AbbVie, Eli Lilly, Janssen, Novartis, Pfizer, and UCB, Consultant of: AbbVie, Amgen, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Eli Lilly, Galapagos, GSK, Janssen, Novartis, Pfizer, and UCB, Grant/research support from: AbbVie, Eli Lilly, GSK, Novartis, Pfizer, and UCB, Philip Helliwell Consultant of: Galapagos, Janssen, and Novartis, Grant/research support from: Abbvie, Janssen, and Pfizer, Christopher T. Ritchlin Consultant of: AbbVie, Amgen, Gilead, Janssen, Lilly, Novartis, Pfizer, UCB Pharma, Grant/research support from: AbbVie, Amgen, UCB Pharma, Alexa Kollmeier Shareholder of: Johnson & Johnson, of which Janssen Research & Development, LLC is a wholly owned subsidiary, Employee of: Janssen Research & Development, LLC, Elizabeth C Hsia Shareholder of: Johnson & Johnson, of which Janssen Research & Development, LLC is a wholly owned subsidiary, Employee of: Janssen Research & Development, LLC, Xie L Xu Shareholder of: Johnson & Johnson, of which Janssen Research & Development is a wholly owned subsidiary, Employee of: Janssen Research & Development, LLC, Shihong Sheng Shareholder of: Johnson & Johnson, of which Janssen Research & Development, LLC is a wholly owned subsidiary, Employee of: Janssen Research & Development, LLC, Yusang Jiang Employee of: Cytel, Inc., providing statistical support (funded by Janssen), Yan Liu Shareholder of: Johnson & Johnson, of which Janssen Research & Development, LLC is a wholly owned subsidiary, Employee of: Janssen Research & Development, LLC, Chenglong Han Shareholder of: Johnson & Johnson, of which Janssen Research & Development, LLC is a wholly owned subsidiary, Employee of: Janssen Research & Development, LLC.