

Background: Patients with lupus nephritis (LN) might be more susceptible to COVID-19 due to the underlying disease, co-morbidities and use of immunosuppressants. We hypothesized that telemedicine (TM) could be a well-accepted mode of health-care delivery minimizing the risk of exposure to the severe acute respiratory syndrome coronavirus 2 ( SARS - CoV - 2 ), while maintaining disease control in these patients.
Objectives: To evaluate the short-term patient satisfaction, compliance, disease control and infection risk of TM compared with standard in-person follow-up (FU) for patients with LN during COVID-19.
Methods: This was a single-center randomized-controlled study. Consecutive patients followed at the LN clinic were randomized to either TM (TM group) or standard FU (SF group) in a 1:1 ratio. Patients in the TM group received scheduled follow-ups via videoconferencing. SF group patients continued conventional in-person outpatient care. The 6-month data were compared.
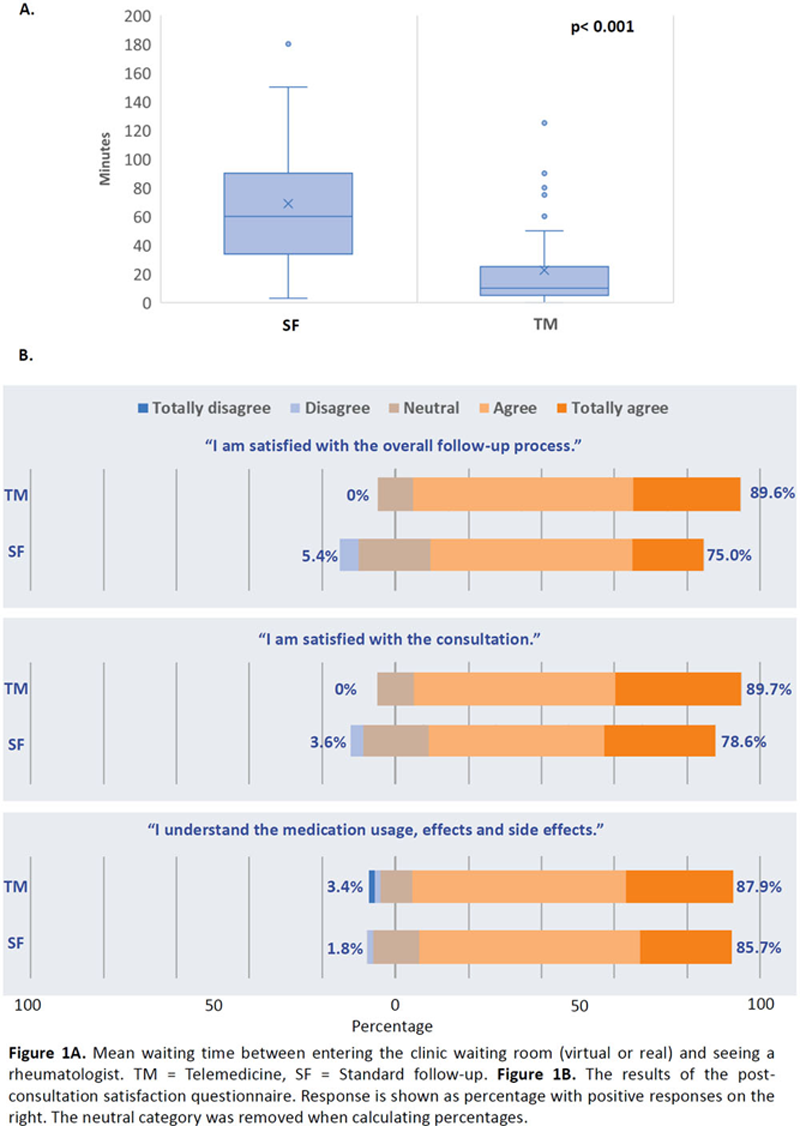
Results: From June to December 2020, 122 patients were randomized (TM: 60, SF: 62) and had attended at least 2 FU visits. There were no baseline differences, including SLEDAI-2k and proportion of patients in lupus low disease activity state (LLDAS), between the 2 groups except a higher physician global assessment score (PGA) in the TM group (mean 0.67±0.69 vs 0.45±0.60, p=0.003) (
Conclusion: TM resulted in better patient satisfaction and could achieve similar disease control in patients with LN in the short-term when compared to standard care.
Baseline clinical data of the recruited patients and comparison between the telemedicine/standard follow-up groups
| Overall (n=122) | Telemedicine group (n=60) | Standard follow-up group (n=62) | P-value | |
| Age in years | 44.4±11.5 | 44.1±11.7 | 44.7±11.5 | 0.779 |
| Gender: Female | 111 (91.0) | 55 (91.7) | 56 (90.3) | 0.796 |
| Disease duration in years | 15.1±9.0 | 16.2±8.7 | 14.0±9.1 | 0.115 |
| Nephritis class III, IV or V | 108 (88.5) | 54 (90.0) | 54 (87.1) | 0.427 |
| 24 hour urine proteinuria in gram | 0.51±0.63 | 0.53±0.60 | 0.50±0.65 | 0.712 |
| Current use of prednisolone | 112 (91.8) | 57 (95.0) | 55 (88.7) | 0.323 |
| Daily prednisolone dose in mg | 5.51±4.21 | 5.69±4.17 | 5.34±4.29 | 0.570 |
| Use of immunosuppressant | 90 (73.8) | 46 (76.7) | 44 (71.0) | 0.474 |
| SLEDAI-2K | 3.65±2.33 | 4.00±2.34 | 3.30±2.29 | 0.097 |
| PGA | 0.56±0.65 | 0.67±0.69 | 0.45±0.60 | 0.003 |
| LLDAS | 78 (63.9) | 36 (60.0) | 42(67.7) | 0.251 |
| Remission | 0 (0) | 0 (0) | 0 (0) | n/a |
| Presence of comorbidity | 87 (71.3) | 40 (66.7) | 47 (75.8) | 0.264 |
| SDI | 0.93±1.15 | 1.08±1.28 | 0.78±0.98 | 0.243 |
| HAQ-DI | 0.23±0.46 | 0.25±0.47 | 0.21±0.44 | 0.571 |
| HADS:
| 6.07±4.12
| 6.20±4.19
| 5.93±4.09
| 0.720
|
Data are reported as mean ± SD or number (%). LLDAS: lupus low disease activity state; SDI: Systemic Lupus International Collaborating Clinics/American College of Rheumatology (SLICC/ACR) Damage Index; HAQ-DI: Health Assessment Questionnaire Disability Index; and HADS: Hospital Anxiety and Depression Scale.
Disclosure of Interests: Ho SO: None declared, Evelyn Chow: None declared, Tena K. Li: None declared, Sze-Lok Lau: None declared, Isaac T. Cheng: None declared, Cheuk-Chun Szeto: None declared, Lai-Shan Tam Grant/research support from: Grants from Novartis and Pfizer.