

Background: Even though corticosteroids and cDMARDs are effective for inducing remission in patients with Adult-Onset Still Disease (AOSD), relapse is common. Hence, maintaining the clinical stability is challenging. Almost all of the patients face side effects because of high dose steroid treatment. Biological DMARDs have been reported to be effective in refractory patients.
Objectives: We aimed to evaluate the patients’ outcomes who were diagnosed with AOSD and treated with at least one bDMARDs in our tertiary center.
Methods: Patients with AOSD who were followed in our clinic between 2007 and 2020 were screened retrospectively. For the diagnosis of AOSD, all of the patients fulfilled Yamaguchi criteria. The demographic characteristics, baseline and post-treatment clinical findings and outcomes were reported.
Results: Twenty-eight patients (21 F, 7 M) were screened (
Comparison of important laboratory findings and the mean steroid dose
| Clinical finding |
At initiation of bDMARD
|
At the last visit
|
| PG-VAS | 9.8 ± 0.8 | 2.3 ± 2.3 |
| ESR (mm/h) | 34,28 ± 33,95 | 18.82 ± 11.60 |
| CRP (mg/l) | 70,76 ± 67,80 | 13.44 ± 27.33 |
| Ferritin (ng/mL) | 1662 ± 1239 | 275.7 ± 381.4 |
| Daily steroid dose (prednisolone, mg/d) | 34.28 ± 26.70 | 5.60 ± 8.60 |
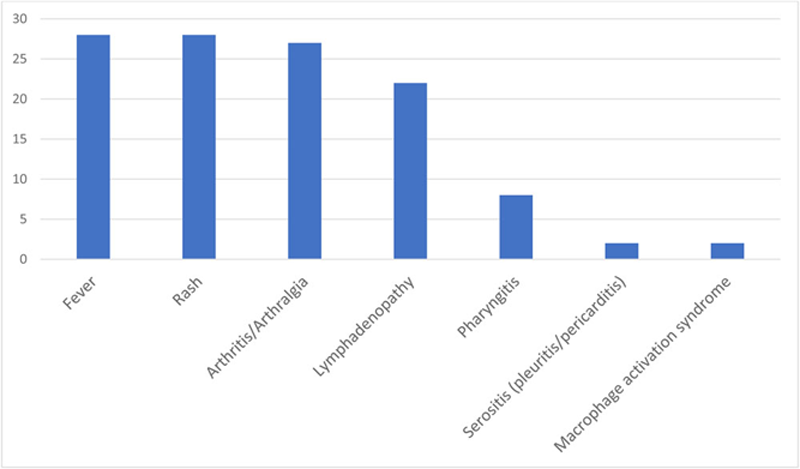
Presenting signs and the symptoms of the patients
bDMARD treatment was terminated in 5 patients due to complete remission (n=2) and side effects (1 of them pneumonia, 1 of them EBER (+) Hodgkin Lymphoma and 1 of them tuberculosis). Six patients experienced local injection site reaction, none of them stopped treatment. Also, one patient died while she was in remission under anakinra treatment with an unknown cause.
Conclusion: The most common presenting symptoms in our cohort were fever and salmon-colored rash. Tocilizumab is an alternative treatment modality in cases with chronic arthritis and IL-1 inhibitors are an alternative for systemic course of disease. bDMARDs, especially IL-1 inhibitors are highly effective for refractory patients with AOSD.
Disclosure of Interests: None declared