

Background: Type I autoimmune pancreatitis (AIP) is a rare form of pancreatitis that belongs to the IgG4-related disease (IgG4-RD) spectrum (1). Autoimmune pancreatitis responds brilliantly to glucocorticoids, but iatrogenic toxicity associated with prolonged steroid therapy represents a relevant clinical concern in the long-term (2). B cell depletion therapy proved to be effective in inducing IgG4-RD remission, yet most evidence are derived from small and heterogenous cohorts.
Objectives: We performed a systematic review and meta-analysis to estimate the remission rate following rituximab-based induction treatment.
Methods: Two reviewers searched MEDLINE, SCOPUS, and EMBASE until December 2020 to identify studies on remission rate and relapse rate of Type I AIP after induction of remission with rituximab. Studies involving three or more patients were included, including the most recent or most numerous cohorts in case of duplicates This study was conducted in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. Pooled effects were calculated using a random-effects model and expressed in terms of pooled remission, relapse and adverse effects (AEs) rates.
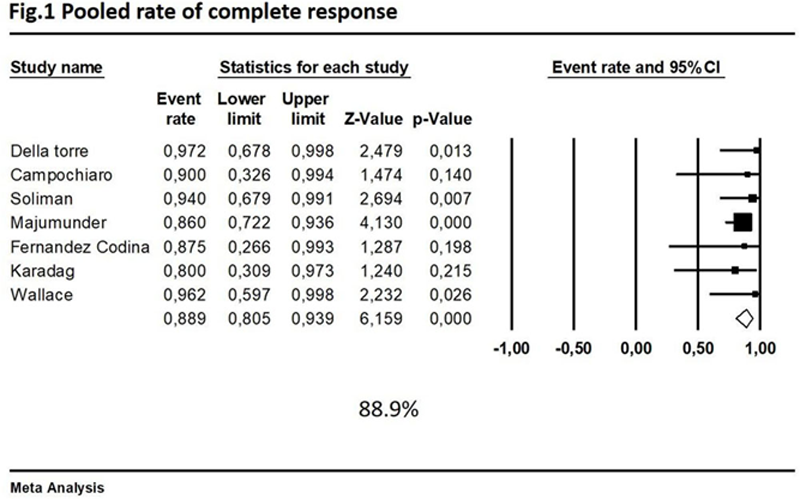
Results: Seven cohort studies met inclusion criteria for the meta-analysis for a total of 101 patients. Reasons for rituximab administration were relapsing disease (63.5%), new diagnosis (18.5%) or glucorticoids intolerance (17.9%). The median follow-up time was 18.5 months. The pooled rate of complete response at 6 months from induction of remission was 88.9% (95% CI 80.5%-93.9%) without heterogeneity (I
2
=0%) (
Conclusion: Treatment of type I AIP with rituximab is associated with high remission rate, a higher relapse rate in presence of OOI and limited AEs. Randomized controlled trials with adequate power are needed to confirm these findings.
REFERENCES:
[1]Lanzillotta M, Mancuso G, Della-Torre E. Advances in the diagnosis and management of IgG4 related disease. BMJ. 2020;369:m1067
[2]Lanzillotta M, Vinge-Holmquist O, Overbeek KA, et al. PrescrAIP: A Pan-European Study on Current Treatment Regimens of Auto-Immune Pancreatitis. Front Med (Lausanne). 2020;7:408
Disclosure of Interests: None declared