

Background: Defects in regulatory T cell (Treg) number and function are associated with autoimmune diseases including SLE. Interleukin (IL)-2 is essential for the development and suppressive function of Treg, and therapies that exploit the ability of IL-2 to expand Treg have shown disease-modifying potential in SLE 1 . However, low-dose IL-2 has a short half-life and narrow selectivity for Treg over conventional CD4+ T cells (Tcon) and natural killer (NK) cells. Efavaleukin alfa is an IL-2 mutein Fc fusion protein; an introduced mutation decreases binding to IL-2Rβ and increases dependence on IL-2Rα (CD25). This preferential binding to the high-affinity IL-2R, constitutively expressed at high levels on Treg, leads to increased cell surface retention and sustained Treg signaling compared with recombinant IL-2. In healthy subjects, a single dose of efavaleukin alfa was well tolerated and led to robust and selective Treg expansion 2 .
Objectives: This final analysis of a phase 1b, double-blind, placebo-controlled, multiple ascending dose study (NCT03451422) reports the safety, tolerability, pharmacokinetics (PK), and pharmacodynamic effects of efavaleukin alfa in patients with SLE.
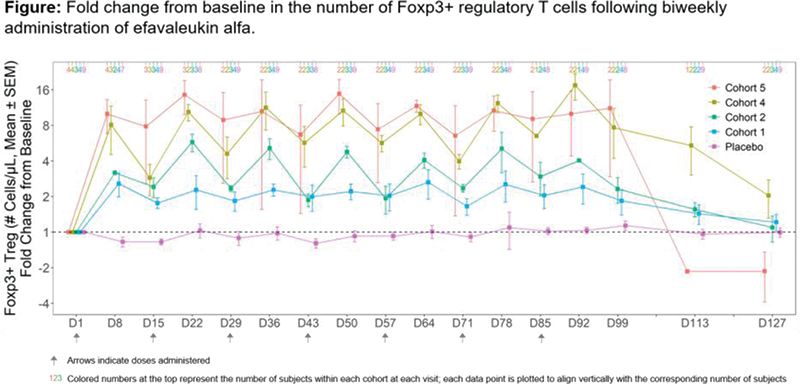
Methods: The study included five ascending dose cohorts (cohort 1=lowest dose; cohort 5=highest dose). A total of 35 patients with SLE (age 24–71 years; 85.7% female; SLE diagnosed using SLICC or ACR criteria with ANA ≥1:80 and/or elevated anti-dsDNA antibodies) were randomized to receive efavaleukin alfa or placebo (5:2 ratio for cohorts 1–3; 3:1 ratio for cohorts 4–5) subcutaneously every 2 weeks (Q2W; cohorts 1, 2, 4, and 5) or every week (QW; cohort 3) in addition to standard of care therapy for a total of 12 weeks, with 6 weeks of follow-up. The primary endpoint was the incidence of treatment-emergent adverse events (TEAEs). Additional endpoints included serum PK of efavaleukin alfa and changes in numbers of Treg, CD4+ Tcon, CD8+ T cells, and NK cells in peripheral blood.
Results: The most commonly reported TEAEs (occurring in ≥25% of efavaleukin alfa-treated subjects) included non-serious, mild or moderate (grade 1–2) injection site reactions. No grade 4 TEAEs or deaths occurred. Two serious AEs were reported in efavaleukin alfa-treated subjects: one event of syncope (grade 3) was observed in cohort 2 and was not considered related to treatment, and one case of eosinophilia (grade 2) was observed in cohort 5 and was considered related to treatment. Efavaleukin alfa PK was generally linear and dose-proportional, with a terminal half-life ranging from 18–30 hours. Peak Foxp3+ Treg expansion was observed at 8 days post-dose, and the magnitude of the peak was generally sustained after multiple QW or Q2W doses. The mean peak increases in Foxp3+ Treg were 14.8-, 17.4-, 5.7-, 2.4-, and 1.1-fold above baseline for efavaleukin alfa Q2W dosing cohorts 5, 4, 2, and 1 and placebo, respectively. At the final study assessment (42 days after the last dose), the mean Treg count was 1.3-fold above baseline (95% CI, 0.9–1.9). Treatment with efavaleukin alfa also expanded CD25 bright Treg (peak 53.8-fold change) and CD31+ recent thymic emigrant (RTE), naïve, and memory Treg subsets. At the highest dose (cohort 5), low-level increases in numbers of CD4+ Tcon (peak 2.3-fold), CD8+ T cells (peak 2.1-fold), and NK cells (peak 2.9-fold) were observed.
Conclusion: Multiple ascending doses of efavaleukin alfa were safe and well tolerated and led to selective and prolonged Treg expansion in SLE patients. Results at the highest dose suggest a plateau in Treg expansion with low-level increases in other IL-2–responsive cells, although interpretation is limited due to small subject numbers. The highest tested dose may be outside the therapeutic window and thus will not be assessed in phase 2 clinical studies. These findings confirm and extend previous results in healthy subjects and support the ongoing phase 2b adaptive randomized controlled trial in patients with SLE.
REFERENCES:
[1]Humrich J. Arthritis Rheumatol. 2016;68 (suppl 10); 2 Tchao N. Blood. 2017;130 (suppl 1).
Acknowledgements: Funding: Amgen Inc
Disclosure of Interests: Nadia Tchao Shareholder of: Amgen Inc., Employee of: Amgen Inc., Nandita Sarkar Shareholder of: Amgen Inc., Employee of: Amgen Inc., Xuguang Hu Shareholder of: Amgen Inc., Employee of: Amgen Inc., Rong Zhang Shareholder of: Amgen Inc., Employee of: Amgen Inc., Cassandra Milmont Shareholder of: Amgen Inc., Employee of: Amgen Inc., Yan Shi Jin Shareholder of: Amgen Inc., Employee of: Amgen Inc., Vincent Chow Shareholder of: Amgen Inc., Employee of: Amgen Inc., Mark Kroenke Shareholder of: Amgen Inc., Employee of: Amgen Inc., Kevin Gorski Shareholder of: Amgen Inc., Employee of: Amgen Inc., Richard Furie Consultant of: Amgen Inc, Grant/research support from: Amgen Inc, Alan Kivitz Shareholder of: Amgen Inc., Gilead, GlaxoSmithKline, Novartis, Pfizer, and Sanofi, Speakers bureau: AbbVie, Celgene, Flexion, Genzyme, GSK, Horizon, Lilly, Merck, Novartis, Pfizer, Sanofi, and UCB, Consultant of: AbbVie, Boehringer Ingelheim, Flexion, Gilead, Janssen, Pfizer, Sanofi, and Sun Pharma, Stanley Cohen Consultant of: Amgen Inc., AbbVie, Eli Lilly, Pfizer, Genentech, and Gilead, Grant/research support from: Amgen Inc., AbbVie, Eli Lilly, Pfizer, Genentech, and Gilead