

Background: Interleukin-6 (IL-6) is elevated in patients with active polymyalgia rheumatica (PMR) and is associated with disease activity, relapse and severity. Clinical trials with IL-6 receptor (IL-6R) inhibitors in PMR showed higher remission rates and reduced glucocorticoid (GC) use vs GC alone. 1-4
Objectives: The SAPHYR study (NCT03600818) assessed the efficacy and safety of sarilumab (SAR), a fully human anti IL-6Rα monoclonal antibody, with a 14 week (wk) GC taper in patients with steroid resistant active PMR who flared on ≥7.5 mg/day prednisone or equivalent.
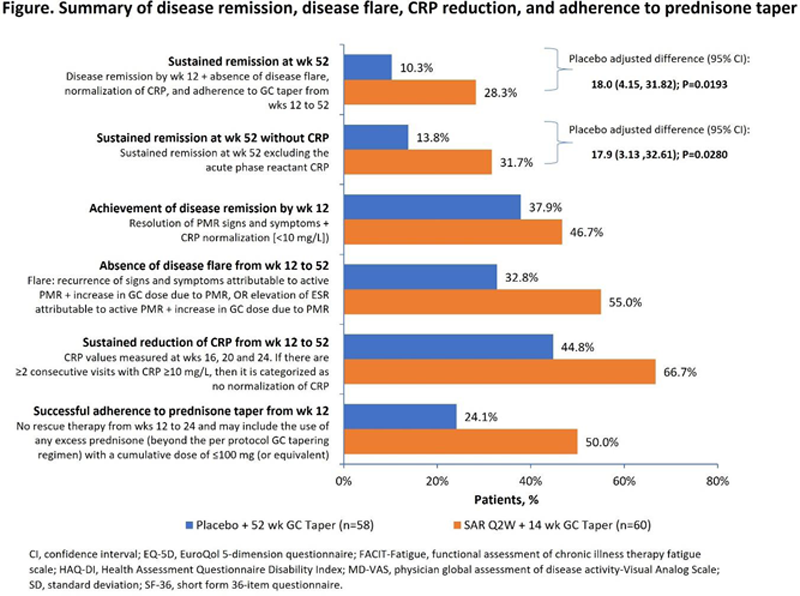
Methods: Patients were randomized (1:1) to 52 wks of treatment with SAR 200 mg every 2 wks (Q2W) + 14 wk GC tapered regimen (SAR arm) OR placebo Q2W + 52 wk GC tapered regimen (comparator arm). The primary endpoint was the proportion of patients achieving sustained remission at wk 52, defined as disease remission by wk 12, absence of disease flare, CRP normalization from wks 12 to 52 and adherence to the per protocol GC taper from wks 12 to 52.
Results: The study was terminated early due to protracted recruitment timelines during the COVID-19 pandemic, resulting in 118 of the intended 280 patients recruited between Oct 2018 and Jul 2020, and 117 were treated (SAR n=59, comparator n=58). The demographics were balanced; patients were primarily female, Caucasian, and a median age of ~70 years (
Demographics and baseline characteristics
| Parameter | SAR + 14 wk GC taper | Placebo + 52 wk GC taper |
|---|---|---|
| (n=60) | (n=58) | |
| Age, median years (range ) | 69 (51–88) | 70 (52–88) |
| Sex (female), n (% ) | 45 (75.0) | 37 (63.8) |
| Race, n (% ) | ||
| Caucasian | 50 (83.3) | 48 (82.8) |
| Asian | 1 (1.7) | 2 (3.4) |
| Not reported | 9 (15.0) | 8 (13.8) |
| PMR duration (diagnosis date to baseline), * median days (range ) | 292 (78–3992) | 310 (66–2784) |
| Any prior disease modifying anti rheumatic drugs, n (% ) | ||
| Methotrexate | 5 (8.3) | 10 (17.2) |
| Leflunomide | 2 (3.3) | 1 (1.7) |
| Azathioprine | 0 | 1 (1.7) |
| Hydroxychloroquine | 1 (1.7) | 1 (1.7) |
| Adalimumab | 1 (1.7) | 0 |
| Tocilizumab | 0 | 1 (1.7) |
| CRP (mg/L), median (range ) | 6.8 (0.5–38.2) | 5.7 (0.1–62.3) |
| Erythrocyte sedimentation rate (mm/h), median (range ) | 25.0 (2.0–115.0) | 22.0 (5.0–85.0) |
*SAR n = 54; comparator n= 50.
Conclusion: SAR + 14 wk GC taper demonstrated significant efficacy vs the comparator arm in steroid refractory PMR patients, including clinically meaningful improvement in quality of life. Safety was consistent with the known safety profile of SAR.
REFERENCES:
[1]Mori 2016;
[2]Akiyama 2020;
[3]Lally 2016,
[4]Devauchelle Pensec 2015
Acknowledgements: Medical writing support was provided by Vijay Kadasi of Sanofi and funded by Sanofi.
Disclosure of Interests: Bhaskar Dasgupta Consultant of: Sanofi, Roche Chugai, Speakers bureau: Roche Chugai, Cipla, Grant/research support from: Sanofi, Roche, Abbvie, Sebastian Unizony Consultant of: Sanofi, Kiniksa, Janssen, Grant/research support from: Genentech, Kenneth J Warrington Paid instructor for: Chemocentryx, Grant/research support from: Eli Lilly, Kiniksa, GSK, Jennifer Sloane Lazar Employee of: Sanofi, Angeliki Giannelou Shareholder of: Regeneron, Employee of: Regeneron, Chad Nivens Shareholder of: Regeneron, Employee of: Regeneron, Bolanle Akinlade Shareholder of: Regeneron, Employee of: Regeneron, Wanling Wong Employee of: Sanofi, Yong Lin Employee of: Sanofi, Frank Buttgereit Consultant of: Sanofi, Horizon Pharma, Roche, Galapagos, Abbvie, Novartis, Grant/research support from: Sanofi, Horizon Pharma, Roche, Galapagos, Abbvie, Novartis, Valerie Devauchelle-Pensec: None declared, Andrea Rubbert-Roth Consultant of: Sanofi, Speakers bureau: Sanofi, Roche, Robert Spiera Consultant of: Sanofi, GSK, Novartis, Chemocentryx, Roche-Genetech, Abbvie, Vera, Grant/research support from: GSK, Chemocentryx, Corbus, Inflarx, Boehringer Ingelheim