

Background: Treat-to-target (T2T) is a generally accepted treatment concept in rheumatology care. It is assumed that T2T could significantly improve systemic lupus erythematosus (SLE) care and the patients’ outcomes. However, T2T has not yet been studied systematically and clinical trials are currently in preparation [1]. Furthermore, the patients’ opinion on T2T has barely been taken into account.
Objectives: As the success of T2T is largely determined by the involvement of patients, it was our aim to investigate the attitude towards, need, and willingness of SLE patients to participate in a T2T study and to identify possible obstacles.
Methods: A questionnaire on T2T, its acceptance, the need and willingness to participate in a T2T trial and possible obstacles for T2T was designed by the authors in cooperation with patient research partner and performed in the Netherlands (NL), Austria (AU), Germany (GE) and Bulgaria (BG). The web-based survey consisted of 13 questions with single and multiple answers and/or free text. After back-and-forth translation from German to Dutch and Bulgarian, it was distributed among members of the patient organizations of NL, GE, AU, BG via newsletter (GE, AU, BG), personal invitation (NL) and a closed Facebook group (BG). Castor Electronic Data Capture (NL) and SoSci-Survey (GE, AU, BG) were used as platforms.
Results: A total of 863 patients (n=316 NL, n=271 GE, n=232 BG, n=44 AU) with self-declared diagnosis of SLE completed the questionnaire. 93.3% were female, 52.2% were 41-60 years old. The disease duration was longer than 10 years in 54.8%, 12.4% had a disease duration of 0-2 years. Regarding the satisfaction with the current health status, 56.2% were somewhat to all the way satisfied, 29.3% were not at all or hardly satisfied. 65.5% were satisfied with their current therapeutic treatment, 14.8% where not at all or hardly satisfied.
48.4% declared being currently in remission, 13% did not know if they were in remission. As most important treatment goal, normalization of quality of life was chosen most frequently (37.4%) followed by prevention of organ damage (24.6%) and the absence of disease activity (22.6 %).
Regarding shared decision making, the majority reported to be somewhat to all the way involved in treatment decisions (62.1%) while 20,7% where hardly or not at all involved.
As most difficult decisions in T2T and shared decision making, respondents named the start of new SLE medication (37.9%) and to change medication while feeling good (39.4%). An increase in the dose of glucocorticoids to reach remission was difficult for 22.7%.
The majority of patients considered research of T2T in SLE useful (55,4% all the way, 29.8% somewhat useful) and 67% would probably or definitely participate in such scientific research. The perceived advantages and disadvantages of T2T are depicted in
Consequences of T2T rated as advantage or disadvantage by SLE patients. SLE systemic lupus erythematosus, T2T treat-to-target.
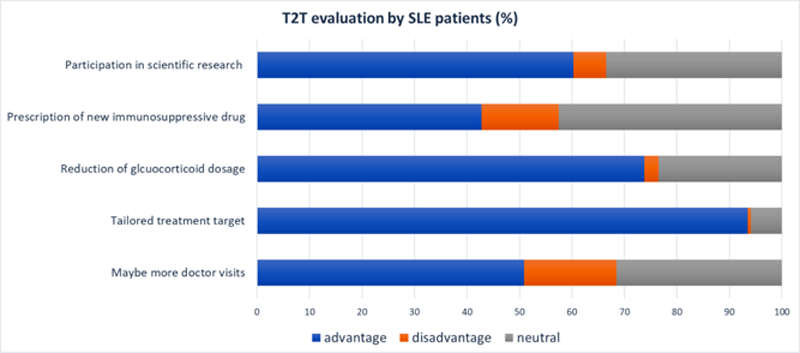
Conclusion: Despite many SLE patients being satisfied with their treatment and health status, T2T is considered an important subject to be studied in clinical trials. Advantages did overweigh possible disadvantages of T2T with the possibility of more doctors’ visits and the prescription of a new drug as biggest disadvantage. Quality of life named as most important treatment goal emphasizes its importance as outcome parameter. Most patients would be willing to participate in a trial about T2T.
REFERENCES:
[1]Mucke J, Kuss O, Brinks R, Schanze S, Schneider M. LUPUS-BEST-treat-to-target in systemic lupus erythematosus: study protocol for a three-armed cluster-randomised trial. Lupus Sci Med 2021; 8(1)
Disclosure of Interests: Johanna Mucke Speakers bureau: AbbVie Deutschland GmbH & Co. KG, Bristol Myers Squibb, Celgene GmbH, Chugai Pharma Germany GmbH, Gilead Sciences Inc., GSK, Janssen-Cilag GmbH, Lilly Deutschlang GmbH, Novartis Pharma GmbH, Consultant of: AbbVie Deutschland GmbH & Co. KG, Amgen, Astra Zeneca, BMS, Celgene GmbH, Gilead Sciences Inc., GlaxoSmithKline, Novartis Pharma GmbH, Lilly Deutschland GmbH, Medac GmbH, Mylan, Grant/research support from: Sanofi Aventis, Daliya Pencheva: None declared, Agner Parra Sanchez Grant/research support from: AstraZeneca, Kyra Kramer: None declared, Matthias Schneider Speakers bureau: MSD, Abbott, AbbVie, Pfizer, GlaxoSmithKline, UCB, Roche, AstraZeneca, Lilly, Janssen-Cilag, Sanofi-Aventis, Chugai, Celgene, Novartis, Boehringer Ingelheim and Bristol-Myers Squibb, Consultant of: MSD, Abbott, AbbVie, Pfizer, GlaxoSmithKline, UCB, Roche, AstraZeneca, Lilly, Janssen-Cilag, Sanofi-Aventis, Chugai, Celgene, Novartis, Boehringer Ingelheim and Bristol-Myers Squibb, Grant/research support from: GSK, UCB, Irene Bultink Speakers bureau: Speaker fees from Eli Lilly, MSD, Amgen, UCB, GSK, Roche, Sanofi Genzyme (outside the submitted work), Consultant of: Consultancy fees from Sanofi Genzyme and AstraZeneca (outside the submitted work)