

Background: Treating-to-target (T2T), a treatment approach focussed on achieving a specific goal by stepwise changes in treatment, is a well-established strategy in various diseases and has been proposed for systemic lupus erythematosus (SLE) 1 . However, the implementation of such strategies has been shown to be difficult in practice with low clinician adherence 2 . Clinical decision support systems (CDSS) are designed to assist health care professionals in their decision-making process 3 . SLE-T2T is an online CDSS tool designed to help rheumatologists to apply a T2T strategy in the management of SLE patients.
Objectives: We aimed to design and develop a first prototype of SLE-T2T, and test its usability for the implementation of a treat-to-target strategy in the management of patients with SLE.
Methods: The creation process was composed of three phases: first, SLE-T2T was conceived as a web-based application with a specific task - to generate appropriate treatment advice based on entered patients’ data. A general sketch of the program was made, and general consensus was achieved with regards to the desired functionalities. In the development phase, a beta version of SLE-T2T was conceived using a free integrated development environment, and based on Javascript, HTML and CSS. After, the system was hosted temporarily on the website of the Amsterdam Rheumatology & Immunology Center 4 , to be accessible for the participants in evaluation phase. Finally, a System Usability Score survey (SUS) 5,6 was chosen as the usability test tool, given its simplicity and advantages; coupled with unstructured feedback about areas of improvement, collected from the participants using the “think aloud” method to determine whether the CDSS was user-friendly, comprehensible, easy-to-deliver, and workflow-oriented.
Results: The SLE-T2T web-based system was developed, consisting of: (1) an input scheme with the set of indexes and scores existing for the measurement of SLE disease activity (cSLEDAI-2K, SLEDAI-2K, PGA score) as well as the patients’ medication; (2) a rule-based interface that collects and processes patients’ data, and (3) an output dashboard with the generated set of recommendations tailored for the patients clinical state and aiming to reach a pre-stablished target of treatment (
Overview of SLE-T2T CDSS tool. a. Architecture of the SLE-T2T app. b. Desktop and mobile example views.
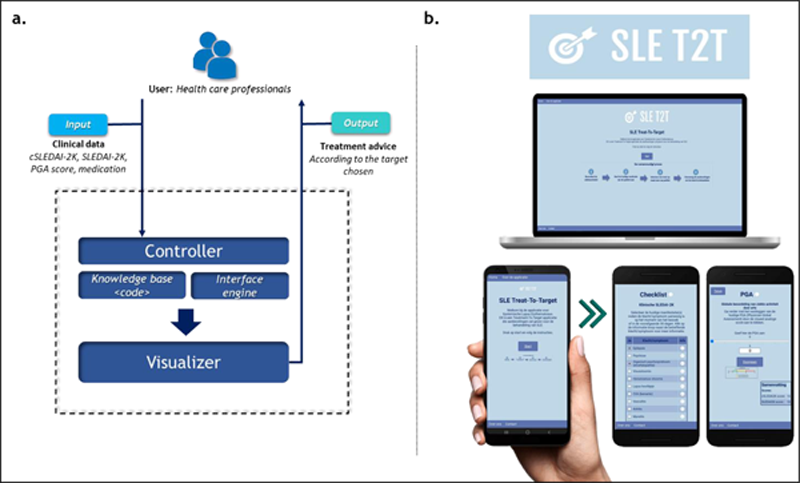
Conclusion: To the best of our knowledge, SLE-T2T is the first eHealth tool to be designed for the management of SLE patients in a treat-to-target context. The SUS score and unstructured feedback showed high acceptance of this digital instrument.
REFERENCES:
[1]van Vollenhoven RF, et al. Treat-to-target in systemic lupus erythematosus: recommendations from an international task force. Ann Rheum Dis. 2014 Jun;73(6):958-67. doi: 10.1136/annrheumdis-2013-205139. Epub 2014 Apr 16.
[2]Lugtenberg M, Zegers-van Schaick JM, Westert GP, Burgers JS. Why don’t physicians adhere to guideline recommendations in practice? An analysis of barriers among Dutch general practitioners. Implement Sci 2009; 4 : 54.
[3]Sutton RT, Pincock D, Baumgart DC, Sadowski DC, Fedorak RN, Kroeker KI. An overview of clinical decision support systems: benefits, risks, and strategies for success. NPJ Digit Med 2020; 3: 17.
[4]
[5]Brooke, J. 1996. “SUS: A “quick and dirty” usability scale”. In Usability evaluation in industry, Edited by: Jordan, P. W., Thomas, B. A. Weerdmeester and McClelland, I. L. 189–194. London: Taylor & Francis.
[6]Lewis, James. (2006). Usability Testing. 10.1002/0470048204.ch49.
Acknowledgements: The work of A.R.P.S. is supported by the European Union’s Horizon 2020 research and innovation programme support for the Amsterdam Rheumatology Centre for Autoimmune Diseases (ARCAID; grant number 847551).
The development phase of SLE-T2T was made possible thanks to the partnership with 2nd year students from the University of Amsterdam Medical Informatics Bachelor’s program.
Disclosure of Interests: Agner Parra Sanchez Grant/research support from: AstraZeneca, Employee of: AstraZeneca, Max G. Grimberg: None declared, Myrthe M.A. Hanssen: None declared, Moon O. Aben: None declared, Elianne E. Jairth: None declared, Prishent S. Dhoeme: None declared, Michel Tsang-A-Sjoe: None declared, Alexandre Voskuyl Speakers bureau: GSK, Consultant of: GSK, AstraZeneca, Roche, Grant/research support from: GSK, UCB, Jan Hendrik Leopold: None declared, Ronald van Vollenhoven Speakers bureau: AbbVie, Galapagos, GSK, Janssen, Pfizer, R-Pharma, UCB, Consultant of: AbbVie, AstraZeneca, Biogen, BMS, Galapagos, Janssen, Miltenyi, Pfizer, UCB, Grant/research support from: BMS, GSK, UCB