

Background: Accurate and practical outcome measures for clinical trials in systemic lupus erythematosus (SLE) are lacking. The SLE Disease Activity Score (SLE-DAS) is a recently validated 17-item instrument, with high accuracy and sensitivity to changes in SLE disease activity. The SLE-DAS definitions of remission and low disease activity (LDA) were newly validated in the clinical setting 1,2 . These definitions may constitute accurate and easy to apply endpoints for SLE trials.
Objectives: (1) To evaluate the ability of SLE-DAS remission and LDA definitions to discriminate drug from placebo in SLE phase 3 trials; (2) To determine if attainment of these SLE-DAS targets are associated with better health-related quality of life (HR-QoL).
Methods: Post-hoc analysis of the merged study population in the BLISS-52 and -76 trials (NCT00424476; NCT00410384) of intravenous belimumab versus placebo for moderate to severe SLE disease activity. We analyzed the British Isles Lupus Assessment Group (BILAG), Physician Global Assessment (PGA), Functional Assessment of Chronic Illness Therapy (FACIT) and 36-Item Short Form Survey (SF-36) trial data. The fulfillment of SLE-DAS remission and LDA definitions were retrospectively assessed from the individual participants’ data. Proportion of patients attaining SLE-DAS Boolean remission (defined as absence of all SLE-DAS clinical items and prednisone ≤5mg/day) and LDA (defined as SLE-DAS≤2.48 and prednisone ≤7.5mg/day), at week 52, was compared between belimumab and placebo arms, using likelihood ratio chi-square test. We further compared the SF-36 physical component summary (PCS) and mental component summary (MCS) and domain scores and the FACIT score between patients attaining SLE-DAS remission vs non-remission and SLE-DAS LDA vs non-LDA, using t-test and Mann-Whitney test.
Results: A total of 1684 SLE patients were included: 562 on placebo, 559 on belimumab 1mg/Kg and 563 on belimumab 10mg/Kg. At week 52, significantly more patients attained SLE-DAS LDA on belimumab 1mg/Kg and 10mg/Kg as compared with placebo (13.0%
vs
17.9%, OR=1.459, p=0.023, and 13.0%
vs
21.7%, OR=1.853, p<0.001, respectively). Likewise, more patients attained SLE-DAS remission on belimumab 10mg/Kg as compared with placebo (10.1%
vs
14.7%, OR= 1.532, p=0.019) (
Attainment of SLE-DAS Boolean remission and LDA at week 52 in BLISS-52 and BLISS-76 trials, according to the treatment groups (n =1684).
| Placebo | Belimumab 1mg/Kg | Belimumab 10mg/Kg | |
|---|---|---|---|
| SLE-DAS remission (n=211) | 10.1% | 12.7%, OR=1.289 (0.89-1.866), p=0.178 | 14.7%, OR 1.532 (1.069-2.195), p=0.019 |
| SLE-DAS LDA (n=295) | 13.0% | 17.9%, OR=1.459 (1.052-2.025), p=0.023 | 21.7%, OR 1.853 (1.349-2.545), p<0.001 |
LDA: Low disease activity; SLE-DAS Boolean remission: absence of all SLE-DAS clinical items and prednisone ≤5mg/day; SLE-DAS LDA: SLE-DAS≤2.48 and prednisone ≤7.5mg/day.
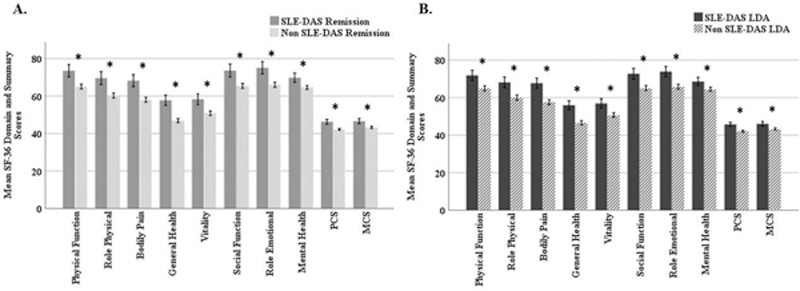
At week 52, patients attaining SLE-DAS remission and SLE-DAS LDA presented higher SF-36 domain and summary scores (all p<0.001) (
Mean SF-36 domain and summary scores at week 52; *p<0.001; MCS, Mental Component Summary; PCS, Physical Component Summary; SF-36, Medical Outcomes Survey Short Form.
Conclusion: The SLE-DAS remission and LDA showed discriminant validity for identifying patients receiving active drug in clinical trials. These treatment targets are associated with better HR-QoL and lower fatigue.
REFERENCES:
[1]Jesus D, et al. Ann Rheum Dis 2021;80:1568-74.
[2]Assuncao H, et al. Rheumatology (Oxford ) 2021;3;keab895.
Acknowledgements: The authors would like to thank GlaxoSmithKline (Uxbridge, UK) for granting access to the data from the BLISS-52 and 76 trials through the Clinical Study Data Request consortium.
Disclosure of Interests: None declared