

Background: Guselkumab (GUS), a human monoclonal antibody targeting the interleukin-23p19 subunit, demonstrated joint and skin efficacy in patients with active psoriatic arthritis (PsA) in the Phase III DISCOVER-1/-2 trials. 1,2 Minimal disease activity (MDA), a multi-domain composite outcome, is a clinically relevant measure of therapeutic response in PsA. 3 However, response dynamics and the effect of individual domains on achieving MDA are not well understood.
Objectives: Characterise response patterns in MDA domains over time and identify potential baseline (BL) response predictors using machine learning.
Methods: Data from bio-naïve patients with active PsA receiving GUS 100 mg every 4 or 8 weeks were pooled across DISCOVER-1/-2. Eligibility criteria are described elsewhere.
1,2
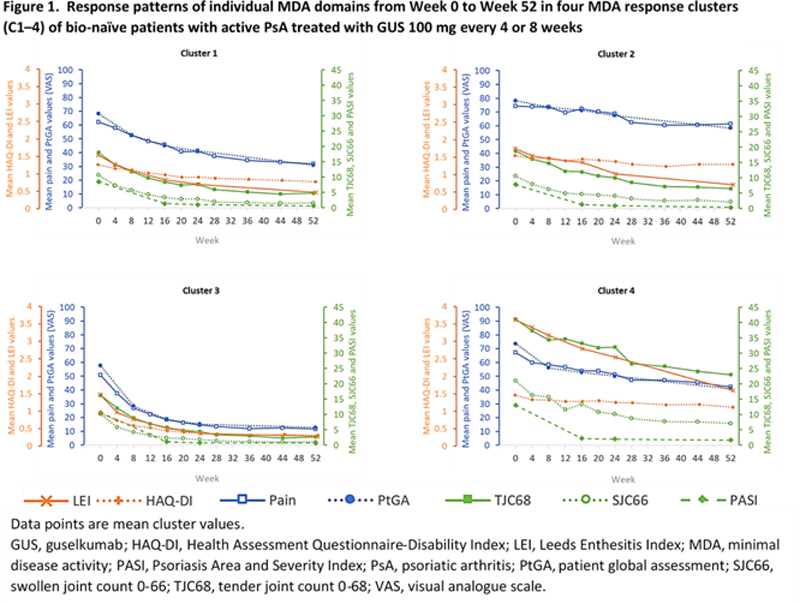
Unsupervised machine learning using the time-series K-means clustering algorithm was performed to identify clusters according to MDA domain responses over 52 weeks. MDA domain thresholds were tender joint count (TJC), swollen joint count (SJC), Psoriasis Area and Severity Index (PASI) and Leeds Enthesitis Index (LEI) each ≤1; patient global assessment (PtGA) visual analogue scale (VAS) ≤20; patient pain VAS ≤15 and Health Assessment Questionnaire-Disability Index (HAQ-DI) ≤0.5. BL characteristics were described for each cluster (
BL characteristics of four MDA response clusters (C1–4) in bio-naïve patients with active PsA treated with GUS 100 mg every 4 or 8 weeks
| Cluster | ||||
|---|---|---|---|---|
| C1 | C2 | C3 | C4 | |
| Patients, N | 201 | 97 | 209 | 64 |
| Age, years | 46.1 ± 12.2 | 45.5 ± 11.5 | 46.1 ± 11.3 | 47.0 ± 11.1 |
| Female, % | 47.8 | 55.7 | 36.4 | 43.8 |
| BMI, kg/m 2 | 29.4 ± 6.3 | 29.2 ± 6.5 | 28.9 ± 5.4 | 28.7 ± 5.7 |
| CRP, mg/dl | 1.8 ± 2.4 | 1.8 ± 1.6 | 1.4 ± 2.0 | 1.9 ± 2.1 |
| PsA disease duration, years | 5.5 ± 5.8 | 5.3 ± 5.7 | 5.2 ± 5.9 | 5.8 ± 5.9 |
| SJC (0–66 ) | 10.7 ± 5.4 | 10.6 ± 5.0 | 10.1 ± 6.2 | 21.1 ± 12.2 |
| TJC (0–68 ) | 18.1 ± 9.6 | 18.8 ± 8.8 | 16.3 ± 10.7 | 41.2 ± 13.3 |
| Spondylitis, % | 28.9 | 26.8 | 29.7 | 42.2 |
| PASI score (0–72 ) | 8.6 ± 8.9 | 7.8 ± 8.0 | 10.6 ± 12.0 | 13.1 ± 14.6 |
| Dactylitis, % | 42.8 | 42.3 | 35.4 | 71.9 |
| Dactylitis score (0–60 ) | 3.7 ± 6.9 | 1.6 ± 2.9 | 2.0 ± 4.4 | 11.7 ± 15.2 |
| Dactylitis count (0–20 ) | 2.1 ± 4.0 | 0.9 ± 1.7 | 1.3 ± 2.8 | 6.0 ± 6.8 |
| Enthesitis, % | 62.2 | 62.9 | 56.9 | 89.1 |
| LEI (0–6 ) | 1.5 ± 1.6 | 1.7 ± 1.8 | 1.5 ± 1.7 | 3.6 ± 2.0 |
| HAQ-DI (0–3 ) | 1.3 ± 0.6 | 1.5 ± 0.5 | 0.9 ± 0.6 | 1.5 ± 0.5 |
| PtGA (0–100 VAS ) | 68.3 ± 17.8 | 78.2 ± 14.1 | 57.6 ± 21.6 | 73.7 ± 15.8 |
| Pain (0–100 VAS ) | 62.1 ± 17.0 | 74.3 ± 13.7 | 50.8 ± 21.3 | 67.2 ± 15.2 |
Data are mean ± standard deviation or %.
CRP, C-reactive protein.
Results: This analysis included 571 of 669 patients receiving GUS and identified four distinct response clusters (C1–4;
Conclusion: Machine learning identified four clusters of GUS-treated PsA patients based on differing response patterns in individual MDA domains. Response types may differ due to BL disease burden, especially in patients with higher pain, PtGA and functional disability scores. These results offer an innovative, complementary approach to identifying treatment response patterns across diverse clusters of bio-naïve patients with PsA, which may facilitate clinical decision-making.
REFERENCES:
[1]Deodhar et al . Lancet 2020; 395: 1115–25.
[2]Mease et al . Lancet 2020; 395: 1126–36.
[3]Coates et al . Ann Rheum Dis 2010; 69: 48–53.
Disclosure of Interests: Alen Zabotti Speakers bureau: AbbVie, Amgen, Janssen, Lilly, Novartis and UCB, Grant/research support from: Novartis, Sarah Ohrndorf Speakers bureau: Sarah Ohrndorf has received speaker fees or travel expense reimbursements from AbbVie, BMS, Janssen, Novartis and Pfizer., William Tillett Speakers bureau:, Consultant of:, Grant/research support from: William Tillett has received research funding, consulting and/or speaker fees from AbbVie, Amgen, Celgene, Janssen, Lilly, MSD, Novartis, Pfizer and UCB., Marlies Neuhold Shareholder of: Johnson & Johnson, Employee of: Janssen, Michel van Speybroeck Shareholder of: Johnson & Johnson, Employee of: Janssen, Elke Theander Shareholder of: Johnson & Johnson, Employee of: Janssen, Christine CONTRE Shareholder of: Johnson & Johnson, Employee of: Janssen, Mohamed Sharaf Shareholder of: Johnson & Johnson, Employee of: Janssen, May Shawi Shareholder of: Johnson & Johnson, Employee of: Janssen, Michelle Perate Shareholder of: Johnson & Johnson, Employee of: Janssen, Alexa Kollmeier Shareholder of: Johnson & Johnson, Employee of: Janssen, Pascal Richette Speakers bureau:, Consultant of: Pascal Richette has received fees from AbbVie, Amgen, Celgene, Janssen, Lilly, MSD, Novartis, Pfizer and UCB.