

Background: The rheumatoid arthritis impact of disease score (RAID) is a patient-reported outcome measure to evaluate the impact of rheumatoid arthritis (RA) on patients’ quality of life (1). While an adapted instrument has been developed for psoriatic arthritis (2), there are no comparable instruments for other inflammatory rheumatic diseases. Since the RAID includes generic questions on pain, functional capacity, fatigue, physical and emotional well-being, sleep disturbances and coping, it could be an informative instrument beyond RA.
Objectives: To analyse the performance of the RAID in ankylosing spondylitis (AS), systemic lupus erythematosus (SLE), polymyalgia rheumatica (PMR), primary Sjögren’s syndrome (pSS), idiopathic inflammatory myositis (IIM), and systemic sclerosis (SSc) compared to RA.
Methods: From 2015 to 2019, a total of 12,398 patients reported the RAID in the National Database of the German Collaborative Arthritis Centres. We calculated the age- and sex-adjusted partial correlation (0.3-0.5 weak, 0.5-0.7 moderate, > 0.7 strong correlation) between the RAID score and five other patient- or physician-reported outcomes, namely patient global (PtGl) health status, PtGl disease activity, physician global (PhGl) disease activity, the World Health Organisation Well-Being Index (WHO-5) and the EuroQoL-5 Dimensions (EQ-5D). Furthermore, for each of the diagnoses the mean difference between the RAID and the other outcomes was compared with the respective differences for RA. The EQ-5D and WHO-5 were rescaled to match the scale of the RAID (range 0-10). General linear regression was used to assess the age- and sex-adjusted effect of each diagnosis on the difference between the RAID and the five other scores with RA as the referent diagnosis. We defined the effect of a diagnosis as clinically relevant if the mean change of difference was at least one unit.
Results: The mean RAID score in RA (3.6) was lower than in AS (4.0) and SSc (3.8) and higher than in SLE, PMR, pSS or IIM (
Characteristics and patient- and physician-reported outcomes in patients with inflammatory rheumatic diseases.
| RA | AS | SLE | PMR | pSS | IIM | SSc | |
|---|---|---|---|---|---|---|---|
| Number of cases | 7826 | 1532 | 1142 | 1105 | 301 | 106 | 386 |
| Female (%) | 74 | 42 | 89 | 64 | 89 | 58 | 76 |
| Age, in years (mean±SD) | 63±14 | 51±14 | 47±15 | 73±8 | 53±16 | 58±14 | 58±14 |
| Disease duration, in years (mean±SD) | 13±11 | 18±13 | 15±10 | 5±6 | 12±9 | 11±8 | 12±10 |
| RAID (mean±SD ) | 3.6±2.3 | 4.0±2.3 | 3.0±2.4 | 3.2±2.3 | 3.4±2.4 | 3.5±2.5 | 3.8±2.3 |
| PtGl health status (mean±SD) | 4.2±2.3 | 4.4±2.2 | 3.6±2.4 | 4.1±2.3 | 4.1±2.3 | 4.2±2.5 | 4.6±2.1 |
| PtGl disease activity (mean±SD) | 3.7±2.4 | 4.0±2.4 | 2.7±2.5 | 3.8±2.7 | 3.5±2.6 | 3.7±2.5 | 4.1±2.3 |
| PhGl disease activity (mean±SD) | 1.8±1.9 | 2.2±1.9 | 1.6±1.3 | 1.0±1.4 | 1.8±1.3 | 1.8±1.6 | 2.2±1.6 |
| EQ-5D (mean±SD) | 0.8±0.2 | 0.8±0.2 | 0.8±0.2 | 0.8±0.2 | 0.8±0.2 | 0.8±0.2 | 0.8±0.2 |
| WHO-5 (mean±SD) | 57±25 | 53±23 | 58±25 | 56±26 | 57±25 | 60±26 | 53±24 |
SD, Standard Deviation; PtGl, Patient Global; PhGl, Physician Global; EuroQoL-5 Dimensions (EQ-5D); World Health Organisation Well-Being Index (WHO-5)
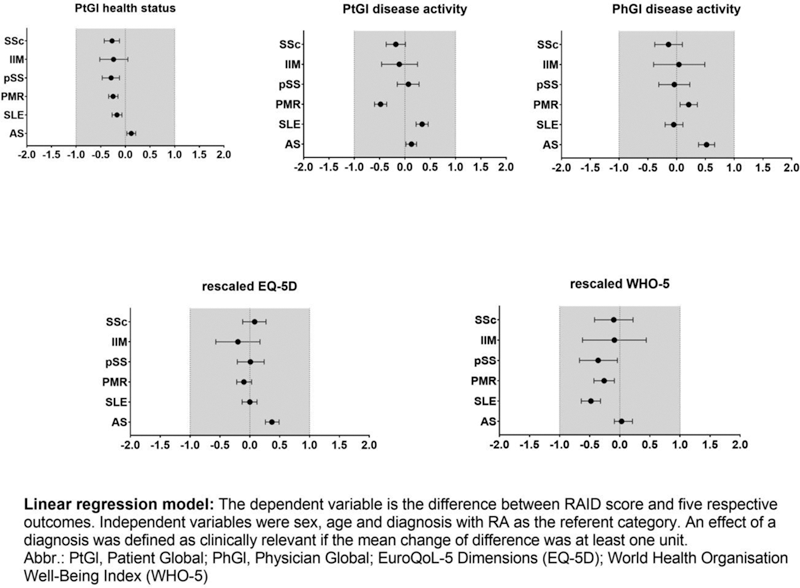
Conclusion: The RAID score performed comparably well across all diagnoses investigated. These findings support the use of the RAID for measuring the impact of disease not only in RA, but also in AS, SLE, PMR, pSS, IIM and SSc.
REFERENCES:
[1]PMID: 21540201
[2]PMID: 24790067
Acknowledgements: We thank all participating rheumatologists and patients for their valuable contributions.
The National Database of the German Collaborative Arthritis centers is supported by the Association of Regional Cooperative Rheumatology Centres, the German Society for Rheumatology and joint contributions to the Rheumatological Training Academy and the German Rheumatism Research Centre by the following members of the Working Group of Corporate Members of the German Society for Rheumatology: AbbVie, AstraZeneca, BMS, GALAPAGOS, GSK, Lilly, Medac, MSD, Pfizer, Sanofi-Aventis and UCB.
Disclosure of Interests: Katja Thiele: None declared, Katinka Albrecht: None declared, Angela Zink: None declared, Martin Aringer: None declared, Kirsten Karberg: None declared, Susanna Spaethling-Mestekemper Speakers bureau: BMS, Gilead, GSK, Janssen, Lilly, Novartis, Pfizer, Sanofi, UCB, Consultant of: Abbvie, Gilead, GSK, UCB, Novartis, Ulrich von Hinueber: None declared, Johanna Callhoff Paid instructor for: Rheumatologische Fortbildungsakademie GmbH, Grant/research support from: Abbvie, AstraZeneca, BMS, GALAPAGOS, GSK, Lilly, Medac, MSD, Pfizer, Sanofi, UCB