

Background: Fatigue, chronic pain, and skin and joint manifestations can be debilitating for people with systemic lupus erythematosus (SLE), and managing these symptoms is vital to safeguard their quality of life (QOL). In addition, oral corticosteroids (OCS) and other immunosuppressants can expose patients with SLE to severe adverse events. However, little is known about the relative importance of SLE treatment outcomes including symptom control or the extent to which treatment risks are tolerated in exchange for this.
Objectives: To quantify the relative importance of different SLE treatment outcomes and the trade-offs patients are willing to make between these outcomes.
Methods: This online discrete choice experiment (DCE) assessed how treatment choices varied with treatment outcomes. Subjects were US adults (≥18 years) with a self-reported diagnosis of moderate-to-severe SLE (≥6 months) who experienced ≥1 flare in the past year and were taking ≥1 SLE treatment. Treatment outcomes included four benefits on: 1) fatigue levels, as in the Functional Assessment of Chronic Illness Therapy (FACIT)–Fatigue, described the degree of tiredness felt by a patient in the previous 7 days, while not experiencing a flare and despite adequate sleep or rest, 2) joint and 3) non-joint pain levels as measured using the Brief Pain Inventory–Short Form (BPI-SF) scale; scores of 2, 5, and 8 correspond to mild, moderate, and severe worst daily pain on a scale of 0 (no pain) to 10 (worst imaginable pain),and 4) skin rash and itch levels, as in SLE-QOL, indicated how much patients were troubled by sore, painful, stingy, or itchy skin in the previous 7 days. Treatment outcomes also included three risks: 1) risk of infections represented by the proportion of patients who might experience an infection during treatment, 2) mild-to-moderate OCS-related side effects requiring no or only self-administered treatment and 3) severe, potentially life-threatening OCS-related side effects requiring long-term outpatient or hospital treatment. An error-component multinomial logit model was used to analyze DCE data.
Results: The DCE survey was completed by 342 subjects with SLE (mean age 39.6 years) who were predominantly female (82%). Most participants were diagnosed with SLE ≥1 year prior (84%) and experienced ≥2 SLE flares in the previous 12 months (90%). Nearly all participants experienced joint pain (92%) and non-joint pain (84%). Treatment preferences were modelled using 3,420 subject-generated observations. Over the ranges tested, more importance was attached to symptom improvement than risk reduction (
Maximum acceptable risk of mild-to-moderate oral corticosteroid side effects
| Attribute levels | Maximum Acceptable Increase in Risk* [95% CI] |
|---|---|
| Fatigue | |
| A little bit | 50.06% [32.41, 67.71] |
| Somewhat | 47.51% [29.92, 65.09] |
| Very much | Reference |
| Skin rash and itch | |
| Not at all | 73.81% [51.51, 96.11] |
| Moderately | 50.84% [32.37, 69.32] |
| Extremely | Reference |
| Joint pain (1-level decrease ) | 15.63% [11.24, 20.02] |
| Non-joint pain (1-level decrease ) | 9.18% [6.06, 12.31] |
| Risk of infections (1% decrease ) | 2.99% [1.53, 4.45] |
| Risk of severe side effects (1% decrease ) | 1.72% [0.97, 2.47] |
*Calculated using an error-component multinomial logit model.
Marginal utilities associated with changes in SLE treatment outcomes
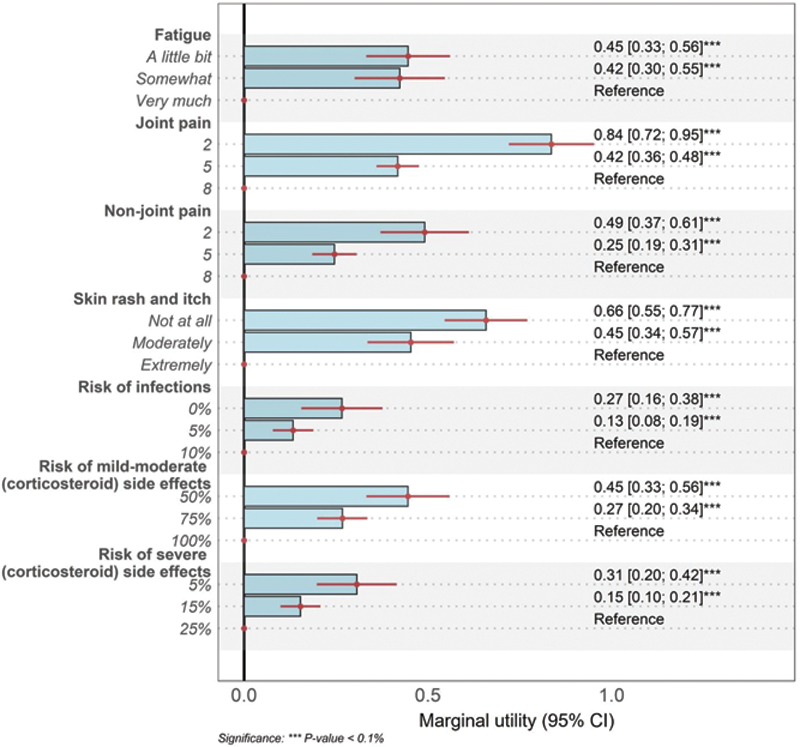
Conclusion: This real-world patient preference assessment provides insights into the relative importance of SLE treatment outcomes and willingness to tolerate SLE treatment risks. These results can guide endpoint prioritization in SLE clinical studies and inform the evaluation of SLE treatments.
Disclosure of Interests: Hannah Collacott Consultant of: Idorsia Pharmaceuticals, Andrea Phillips-Beyer Consultant of: Idorsia Pharmaceuticals, Nicolas Krucien Consultant of: Idorsia Pharmaceuticals, Kevin Marsh Consultant of: Idorsia Pharmaceuticals, Bruno Flamion Employee of: Idorsia Pharmaceuticals