

Background: Accurate and practical outcome measures for clinical trials in systemic lupus erythematosus (SLE) are lacking. The SLE Disease Activity Score (SLE-DAS) is a recently validated 17-item instrument, with high accuracy and sensitivity to changes in SLE disease activity. The SLE-DAS definitions of remission and low disease activity (LDA) were newly validated against disease activity physician-applied measures in the clinical setting [1, 2]. Criterion validity of SLE-DAS for Patient Reported Outcomes, namely health-related quality of life (HR-QoL) and fatigue needs to be assessed.
Objectives: To evaluate if the attainment of SLE-DAS remission and LDA states is associated with improvements in HR-QoL and fatigue.
Methods: Post-hoc analysis of the merged study population in the BLISS-52 and -76 trials (NCT00424476; NCT00410384) of intravenous belimumab versus placebo for moderate to severe SLE disease activity. We analysed the Functional Assessment of Chronic Illness Therapy (FACIT) and 36-Item Short Form Survey (SF-36) trial data. Fulfillment of SLE-DAS remission (defined as absence of all SLE-DAS clinical items and prednisone ≤5mg/day) and LDA (defined as SLE-DAS≤2.48 and prednisone ≤7.5mg/day) definitions were retrospectively assessed from the individual participants’ data. Mean changes from study baseline to week 52 in FACIT and SF-36 physical component summary (PCS) and mental component summary (MCS) and domain scores were compared between patients attaining at week 52 the SLE-DAS remission vs non-remission and the SLE-DAS LDA vs non-LDA using multivariate regression analysis adjusted for baseline scores.
Results: A total of 1684 SLE patients were included. Few patients were in SLE-DAS remission (0.5%) and LDA (0.8%) at study entry. At week 52, 12.5% patients attained SLE-DAS remission and 17.5% attained SLE-DAS LDA. Mean improvements in SF-36 PCS and MCS scores were greater in patients that attained SLE-DAS remission
vs
non-remission (5.4
vs
3.4, and 4.6
vs
2.7, respectively; multivariate p<0.005 for both) and SLE-DAS LDA
vs
non-LDA (5.0
vs
3.4 and 4.6
vs
2.6, respectively; multivariate p<0.005 for both), at week 52 (
Mean changes in SF-36 domains and summary scores from baseline to week 52. #p<0.005; *p<0.001; MICD, Minimum Clinically Important Difference; MCS, Mental Component Summary; PCS, Physical Component Summary; SF-36, Medical Outcomes Survey Short Form; SLE-DAS, Systemic Lupus Erythematosus Disease Activity Score.
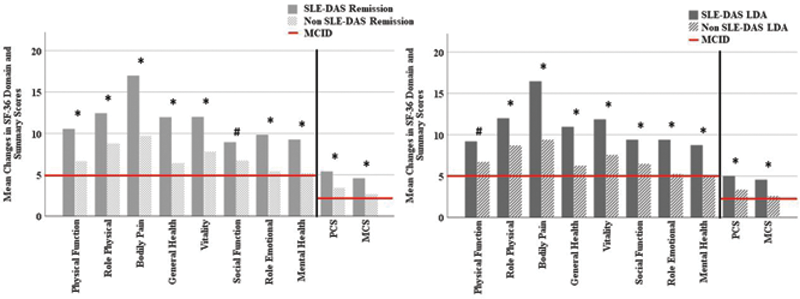
Additionally, mean improvements in FACIT scores were higher in SLE-DAS remission than non-remission (6.3 vs 3.6, multivariate p<0.001) and in SLE-DAS LDA than non-LDA (5.9 vs 3.6, multivariate p<0.001), and exceeded the MCID of 4 points.
Conclusion: Attainment of SLE-DAS remission and LDA is associated with meaningful improvement in HR-QoL and fatigue.
REFERENCES:
[1]Jesus D, et al. Systemic Lupus Erythematosus Disease Activity Score (SLE-DAS) enables accurate and user-friendly definitions of clinical remission and categories of disease activity. Ann Rheum Dis 2021;80:1568-74.
[2]Assunção H, et al. Definition of Low Disease Activity State based on the SLE-DAS: Derivation and validation in a multicentre real-life cohort. Rheumatology (Oxford ) 2021;3;keab895.
Acknowledgements: The authors would like to thank GlaxoSmithKline (Uxbridge, UK) for granting access to the data from the BLISS-52 and 76 trials through the Clinical Study Data Request consortium.
Disclosure of Interests: None declared