

Background: Voclosporin (VCS), a novel calcineurin inhibitor, was approved in the US in January 2021 for the treatment of adult patients with active lupus nephritis (LN) in combination with background immunosuppressive therapy. The Phase 3 AURORA 1 study showed that the addition of VCS to mycophenolate mofetil (MMF) and low-dose steroids in patients with LN significantly increased rates of complete renal response at 52 weeks.
Objectives: Here we report the results of the completed continuation study, AURORA 2, which assessed the long-term safety and tolerability of VCS compared to placebo in patients with LN receiving treatment for an additional 24 months following completion of the AURORA 1 study
Methods: Key inclusion criteria for the parent AURORA 1 study included a diagnosis of biopsy-proven active LN (Class III, IV, or V ± III/IV), proteinuria ≥1.5 mg/mg (≥2 mg/mg for Class V) and estimated glomerular filtration rate (eGFR) >45 mL/min/1.73 m 2 . Patients who completed AURORA 1 and who elected and were eligible to enter AURORA 2 continued on the same blinded therapy as at the end of AURORA 1 (either VCS or placebo twice daily in combination with MMF and low-dose steroids). Safety and tolerability were monitored, and eGFR, serum creatinine (SCr), and urine protein creatinine ratio (UPCR) were also assessed.
Results: In total, 116 and 100 patients in the VCS and control arms enrolled in AURORA 2, with 92 (79.3%) and 73 (73.0%) patients in each respective arm receiving treatment to the end of AURORA 2. There were no unexpected safety signals in the VCS arm compared to control, with similar rates of serious adverse events reported in both arms (VCS [18.1%] vs. control [23.0%];
Overall Summary of Adverse Events
| Control(n=100) | Voclosporin(n=116) | |
|---|---|---|
| n (%) | n (%) | |
| Any AE | 80 (80.0) | 100 (86.2) |
| Renal Impairment | 2 (2.0) | 4 (3.4) |
| Acute Kidney Injury | 0 | 0 |
| Treatment-related AE | 21 (21.0) | 28 (24.1) |
| Serious AE | 23 (23.0) | 21 (18.1) |
| Serious Treatment-related AE | 2 (2.0) | 1 (0.9) |
| AE Leading to Study Drug Discontinuation | 17 (17.0) | 11 (9.5) |
| Death | 4 (4.0) | 0 |
| Treatment-related Death | 0 | 0 |
LS Mean eGFR over Time
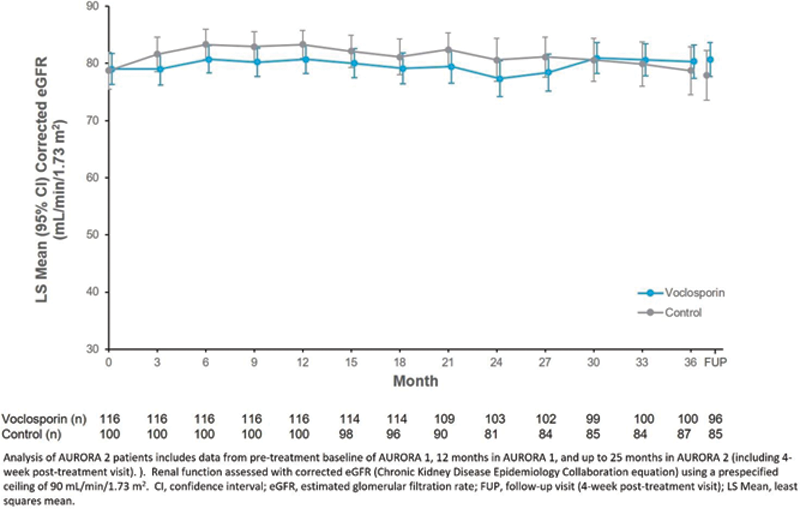
Conclusion: Voclosporin was well-tolerated over 3 years of treatment with no unexpected safety signals detected. Further, eGFR remained stable throughout the study period, and the significant and meaningful reductions in proteinuria achieved in AURORA 1 were maintained. These data provide evidence of a long-term treatment benefit of VCS in patients with LN.
Includes adverse events starting on or after the first dose of study drug in AURORA 2 up to 30 days after the last dose and all events of death reported during study follow-up. Adverse events were aggregated by System Organ Class and Preferred Term and coded using Medical Dictionary for Regulatory Activities (MedDRA) Version 20.0. AE, adverse event.
Disclosure of Interests: Amit Saxena Speakers bureau: Aurinia Pharmaceuticals Inc., Consultant of: Aurinia Pharmaceuticals Inc., Y.K. Onno Teng Consultant of: Aurinia Pharmaceuticals Inc., Chris Collins Shareholder of: Aurinia Pharmaceuticals, Employee of: Aurinia Pharmaceuticals, Nicole England Shareholder of: Aurinia Pharmaceuticals, Employee of: Aurinia Pharmaceuticals, Henry Leher Shareholder of: Aurinia Pharmaceuticals, Employee of: Aurinia Pharmaceuticals