

Background: The first vaccine against SARS-CoV-2 was approved in December 2020. Immunogenicity of SARS-CoV2 vaccines in patients with immune-mediated inflammatory disease (IMID) have so far been evaluated in the 2-6 weeks following complete vaccination and risk groups for poor early vaccine response have been identified leading to specific vaccination recommendations. However, data on the long-term course and persistence of vaccine response in IMID patients, as well as the outcomes of the specific recommendations are lacking.
Objectives: To evaluate the long-term course of humoral response to SARS-CoV-2 vaccination in a large prospective cohort of IMID patients and non-IMID controls with a follow-up duration of up-to to 10 months after the first vaccine dose.
Methods: We have initiated a prospective dynamic cohort of IMID patients and healthy controls in February 2020 to monitor immune response to SARS-CoV-2 and respiratory infections including COVID-19 (1). Participants who contributed data starting from the 4 weeks before their first vaccination onwards were included in this analysis. Antibodies against SARS-CoV-2 spike protein were quantified with an ELISA from Euroimmun (Lübeck, Germany) with an optical density cutoff of 0.8. We fitted linear mixed-effect models for log-transformed antibody levels using time splines with adjustment for age and sex. Marginal mean antibody levels with 95% confidence intervals (CI) were estimated at selected time points for IMID patients and controls with double vaccination. We descriptively analyzed the observed antibody levels over time in cohort participants receiving two vaccinations vs. three vaccinations.
Results: Among 5076 cohort participants, 3147 IMID patients and healthy controls (mean (SD) age 49 (16)) provided 4756 samples for this analysis between December 2020 and 2021, with a median (IQR) 28 (14-31) weeks of follow-up after the first vaccination (
Participant characteristics and antibody levels
| Healthy controls | IMID | |
|---|---|---|
| N | 1199 | 1948 |
| Age, mean (SD) | 40.8 (13.5) | 54.3 (14.8) |
| Follow-up, weeks, median (IQR) | 31.1 (23.8-36.6) | 19.6 (12.3-26.6) |
| Follow-up range, weeks, | 1.6-46.1 | 1.7-46.3 |
| Sex, n(%) | ||
| Female | 554 (46.2) | 1136 (58.3) |
| Vaccine intervals, ´median (IQR) | ||
| 1 st to 2 nd dose | 4.6 (3.0-6.0) | 6.0 (5.0-6.1) |
| 2 nd to 3 rd dose | 29.6 (26.9-36.4) | 19.9 (10.0-26.1) |
| Diagnosis, n (%) | ||
| Spondyloarthritis | - | 713 (36.6) |
| Rheumatoid arthritis | - | 489 (25.1) |
| Autoimmune disease, systemic+ | - | 420 (21.5) |
| Inflammatory bowel disease | - | 219 (11.2) |
| Psoriasis | - | 107 (5.5) |
| Mean* antibody levels after 1 st dose | ||
| Week-8 | 4.16 (3.89 to 4.45) | 2.97 (2.83 to 3.12) |
| Week-16 | 8.39 (7.81 to 9.02) | 5.04 (4.81 to 5.28) |
| Week-32 | 5.02 (4.73 to 5.33) | 2.52 (2.32 to 2.74) |
| Week-40 | 2.14 (1.95 to 2.35) | 1.01 (0.86 to 1.19) |
+ Systemic lupus, systemic sclerosis, Sjögren’s syndrome, vasculitis
* Estimated marginal means adjusted for age and sex.
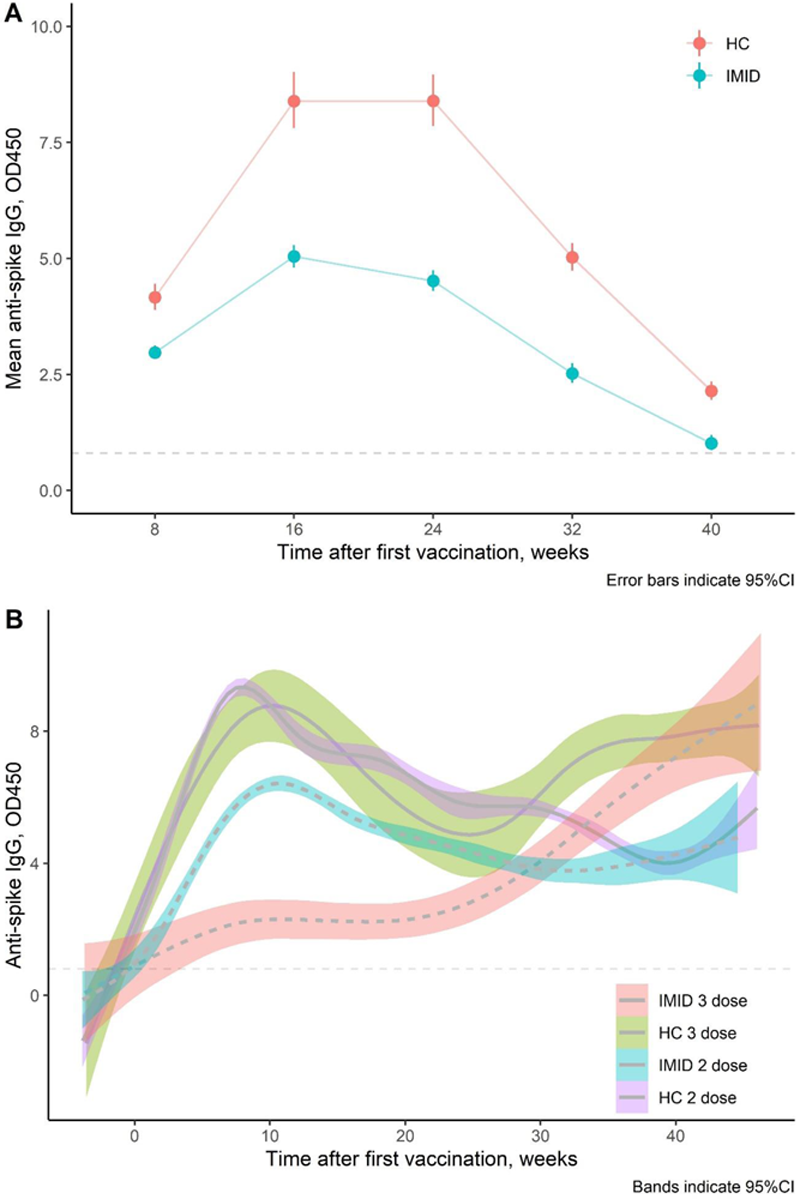
Conclusion: Humoral response to vaccination against SARS-CoV-2 was weaker in IMID patients compared to controls at all time points after the first vaccine dose and practically disappeared after 1 year. IMID patients can still achieve a good antibody response with a third dose even after a weak response with two doses.
REFERENCES:
[1]Simon D et al Nat Commun 2020
Disclosure of Interests: None declared