

Background: Recently, the disease activity of rheumatoid arthritis (RA) was improved due to the ‘treat-to-target’ strategy. However, some patients remain various symptoms despite recommended treatment was performed. Then, the term of ‘difficult-to-treat RA (D2TRA)’ is widely recognized. It is unknown how the difference of type of biological disease-modifying anti rheumatic dugs (bDMARDs)/Janus kinase inhibitor (JAKi) will affect clinical efficacy in patients with D2TRA. Moreover, the risk factor to inhibit the clinical response in patients with D2TRA is unknown.
Objectives: The aim of this study was to evaluate the treatment response in patients with D2TRA who were treated with interleukin 6 receptor inhibitor (IL-6Ri), abatacept and JAKi.
Methods: This study used the multicenter database included 673 RA patients treated with bDMARDs/JAKi (tocilizumab 240, sarilumab 67, abatacept 146, tofacitinib 101, baricitinib 83, upadacitinib 20, peficitinib 14, filgotinib 2). Two hundred forty-two patients were treated as first line bDMARDs/JAKi (IL-6Ri 117, abatacept 63, JAKi 62), 211 patients were treated as second line bDMARDs/JAKi (IL-6Ri 117, abatacept 37, JAKi 57), 220 patients were treated as third and more bDMARDs/JAKi. In these 220 patients, 82 patients did not meet D2TRA criteria (IL-6Ri 42, abatacept 15, JAKi 25) and 138 patients met D2TRA criteria (IL-6Ri 31, abatacept 31, JAKi 76). In all patients, we analyzed 138 patients with D2TRA (113 female, mean age was 63.1 ± 13.7 years). Drug retention rate and effectiveness of bDMARDs/JAKi in patients with D2TRA were evaluated for 24 weeks. Multivariate linear regression analysis was performed to clarify the risk factors to inhibit the clinical response.
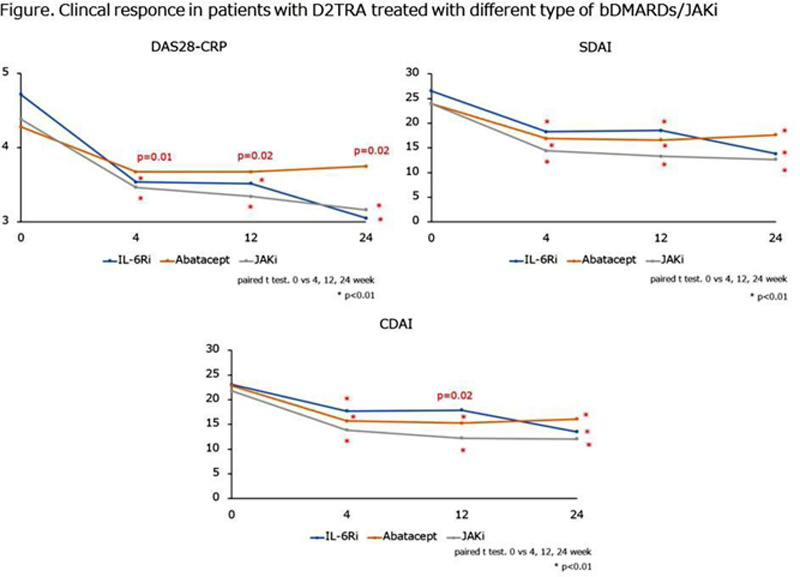
Results: Drug retention rate of patients with D2TRA at 24 weeks was 67.7% in IL-6Ri group, 74.2% in abatacept group, 61.8% in JAKi group. Drug retention rate in patients with D2TRA was not different between groups (IL-6Ri vs abatacept: p=0.86, IL-6Ri vs JAKi group: p=0.39, abatacept vs JAKi group: p=0.33). DAS28-CRP at 4, 12, 24 weeks decreased in all group (
Multivariate linear regression analysis of risk factor to inhibit the clinical response in patients with D2TRA.
| β | 95% CI | p | |
|---|---|---|---|
| Age (years) | -0.037 | -0.025, 0.017 | 0.74 |
| male | -0.047 | -0.788, 0.486 | 0.64 |
| Disease durations (years) | -0.048 | -0.028, 0.017 | 0.63 |
| RF (IU/ml) | -0.082 | -0.0004, 0.0002 | 0.41 |
| Anti CCP antibody (U/ml) | 0.111 | -0.0005, 0.002 | 0.26 |
| DAS28-CRP | -0.063 | -0.265, 0.142 | 0.55 |
| HAQ | 0.279 | 0.059, 0.717 | 0.02 |
| MTX (mg/day) | 0.136 | -0.018, 0.081 | 0.21 |
| Glucocorticoid dose (mg/day) | 0.669 | 0.174, 0.324 | < 0.01 |
| Type of bDMARDs/JAKi | -0.088 | -0.415, 0.151 | 0.36 |
Conclusion: Drug retention rate and clinical efficacy of D2TRA patients were not different among IL-6Ri, abatacept and JAKi. DT2RA patient with functional disorder and high dosage of glucocorticoid were risk factor to inhibit the clinical response.
Disclosure of Interests: None declared