

Background: Tofacitinib is an oral JAK inhibitor for the treatment of adults with ankylosing spondylitis (AS).
Objectives: To describe the tofacitinib safety profile from an integrated analysis of randomised controlled trials (RCTs) in patients (pts) with active AS.
Methods: Pooled data from Phase (P)2 (NCT01786668) and P3 (NCT03502616) RCTs in pts with AS were analysed in 3 cohorts (
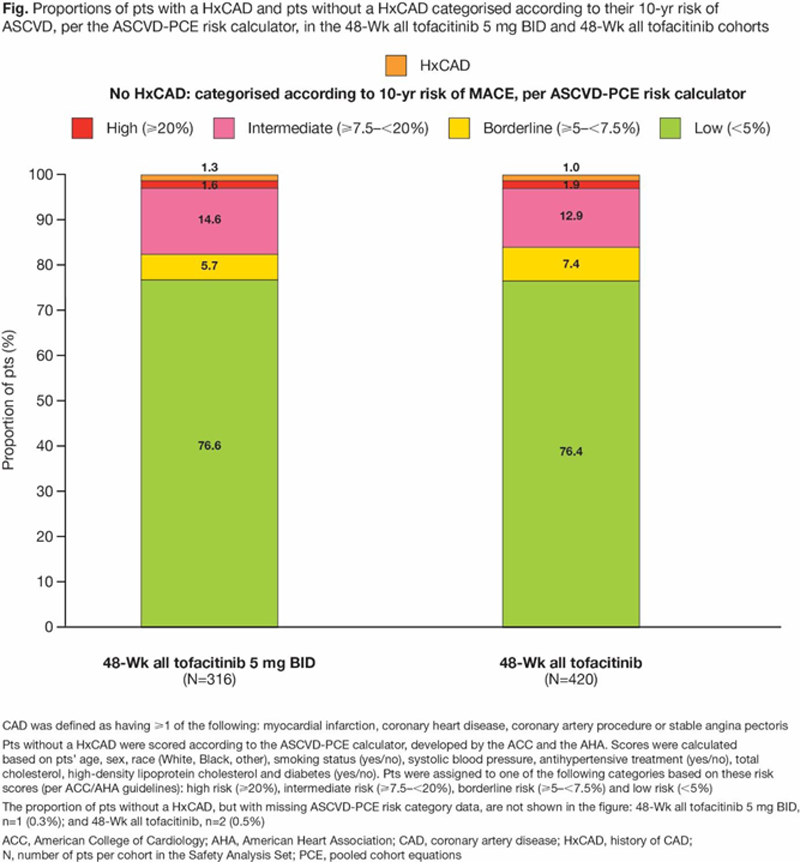
Results: At BL, most pts (>76%) in the 48-Wk tofacitinib cohorts had <5% (low) 10-yr ASCVD risk (
AEs and AEs of special interest
| 16-Wk PBO-controlled cohort | 48-Wk tofacitinib cohorts | |||
|---|---|---|---|---|
| Tofacitinib 5 mg BID N=185 | PBO N=187 | 48-Wk all tofacitinib 5 mg BID N=316 | 48-Wk all tofacitinib N=420 | |
| AE, n (%), IR [95% CI per 100 pt-yrs] | ||||
| Serious AE | 3 (1.6) 5.28 [0.00, 11.25] | 2 (1.1) 3.56 [0.00, 8.49] | 8 (2.5) 3.49 [1.51, 6.87] | 9 (2.1) 3.45 [1.58, 6.55] |
| Discontinuation due to AEs | 4 (2.2) 7.04 [0.14, 13.94] | 4 (2.1) 7.10 [0.14, 14.05] | 11 (3.5) 4.77 [2.38, 8.54] | 12 (2.9) 4.58 [2.37, 8.00] |
| SI | 1 (0.5) 1.77 [0.00, 5.89] | 0 0.00 [0.00, 3.31] | 1 (0.3) 0.43 [0.01, 2.41] | 1 (0.2) 0.38 [0.01, 2.12] |
| HZ | 0 0.00 [0.00, 3.28] | 0 0.00 [0.00, 3.31] | 5 (1.6) 2.18 [0.71, 5.08] | 7 (1.7) 2.68 [1.08, 5.53] |
| All-cause mortality | 0 0.00 [0.00, 3.28] | 0 0.00 [0.00, 3.31] | 0 0.00 [0.00, 1.59] | 0 0.00 [0.00, 1.40] |
| Malignancies excluding NMSC | 0 0.00 [0.00, 3.28] | 0 0.00 [0.00, 3.31] | 0 0.00 [0.00, 1.59] | 0 0.00 [0.00, 1.40] |
| Major adverse CV event | 0 0.00 [0.00, 3.28] | 0 0.00 [0.00, 3.31] | 0 0.00 [0.00, 1.59] | 0 0.00 [0.00, 1.40] |
| Venous thromboembolism | 0 0.00 [0.00, 3.28] | 0 0.00 [0.00, 3.31] | 0 0.00 [0.00, 1.59] | 0 0.00 [0.00, 1.40] |
CI, confidence interval; n, number of pts with event within 28-day risk period
Conclusion: Tofacitinib 5 mg BID was well tolerated over 48 Wks in pts with AS, and safety was consistent with the established safety profile of tofacitinib.
Acknowledgements: Study sponsored by Pfizer Inc. Medical writing support was provided by Jennifer Arnold, CMC Connect, and funded by Pfizer Inc.
Disclosure of Interests: Atul Deodhar Consultant of: AbbVie, Amgen, Aurinia, Boehringer Ingelheim, Bristol-Myers Squibb, Celegene, Eli Lilly, GlaxoSmithKline, Janssen, MoonLake, Novartis, Pfizer Inc and UCB, Grant/research support from: AbbVie, Eli Lilly, GlaxoSmithKline, Novartis, Pfizer Inc and UCB, Servet Akar Speakers bureau: AbbVie, Amgen, Eli Lilly, MSD, Novartis, Pfizer Inc and UCB, Consultant of: AbbVie, Amgen, Eli Lilly, MSD, Novartis, Pfizer Inc and UCB, Grant/research support from: Pfizer Inc, Jeffrey Curtis Consultant of: AbbVie, Amgen, Bristol-Myers Squibb, CorEvitas, LLC (formerly Corrona, LLC), Eli Lilly, Janssen, Myriad, Pfizer Inc, Radius, Roche and UCB, Grant/research support from: AbbVie, Amgen, Bristol-Myers Squibb, CorEvitas, LLC (formerly Corrona, LLC), Eli Lilly, Janssen, Myriad, Pfizer Inc, Radius, Roche and UCB, Bassel Zorkany Consultant of: AbbVie, Amgen, Bristol-Myers Squibb, Eva, Eli Lilly, Hekma, Janssen, MSD, New Bridge, Novartis, Pfizer Inc, Roche, Sanofi-Aventis and Servier, Grant/research support from: AbbVie, Amgen, Bristol-Myers Squibb, Eva, Eli Lilly, Hekma, Janssen, MSD, New Bridge, Novartis, Pfizer Inc, Roche, Sanofi-Aventis and Servier, Marina Magrey Consultant of: AbbVie, Eli Lilly, Novartis, Pfizer Inc and UCB, Grant/research support from: AbbVie and UCB, Cunshan Wang Shareholder of: Pfizer Inc, Employee of: Pfizer Inc, Joseph Wu Shareholder of: Pfizer Inc, Employee of: Pfizer Inc, Solomon B Makgoeng Shareholder of: Pfizer Inc, Employee of: Pfizer Inc, Ivana Vranic Shareholder of: Pfizer Inc, Employee of: Pfizer Ltd, Sujatha Menon Shareholder of: Pfizer Inc, Employee of: Pfizer Inc, Dona Fleishaker Shareholder of: Pfizer Inc, Employee of: Pfizer Inc, Annette Diehl Shareholder of: Pfizer Inc, Employee of: Pfizer Inc, Lara Fallon Shareholder of: Pfizer Inc, Employee of: Pfizer Inc, Arne Yndestad Shareholder of: Pfizer Inc, Employee of: Pfizer Inc, Robert B.M. Landewé Consultant of: AbbVie, AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Galapagos NV, Novartis, Pfizer Inc and UCB