

Background: Guselkumab (GUS), a selective IL-23 inhibitor, is efficacious in treating bio-naïve and TNFi-experienced active PsA patients (pts). 1.2 In the COSMOS study of active PsA pts with lack of efficacy/intolerance, i.e., inadequate response (IR), to 1-2 TNFi, GUS demonstrated significantly greater response rates and mean improvements in PsA signs and symptoms vs. placebo (PBO) at Week (W) 24. 3
Objectives: Evaluate baseline (BL) serum levels of pro-inflammatory biomarkers (CRP, serum amyloid A [SAA], TNFα, IFNɣ, IL-6, IL-10, IL-17F, IL-17A, IL-22) and their relationship to BL disease activity, GUS treatment (tx), and clinical response in COSMOS TNFi-IR pts.
Methods: TNFi-IR pts ≥18 yrs with active PsA (≥3 swollen & ≥3 tender joint counts [SJC/TJC]) were randomized 2:1 to GUS 100 mg every 8 W (Q8W) through W44 or PBO with early escape (W16) or crossover (W24) to GUS Q8W. Samples for serum biomarker analyses, collected at W0, 4, 16, 24, and 48 from consenting pts, were compared with healthy controls (HC; independent of COSMOS). Associations between early biomarker changes and BL disease activity, GUS tx, and clinical response at W24 were assessed.
Results: Among 285 COSMOS pts, 50/95 PBO and 100/190 GUS pts had available biomarker data. BL characteristics of the biomarker cohort were similar to the overall COSMOS population and well balanced across tx arms. At BL, levels of TNFα, IFNɣ, IL-6, IL-10, IL-17A, IL-17F, and IL-22 were significantly upregulated in TNFi-IR pts vs. HC (
Select Serum Biomarkers at BL in TNFi-IR pts vs. HC▫
| Biomarker, pg/mL | HC N=24 | TNFi-IR N=150 | Fold difference | p-value |
|---|---|---|---|---|
| CRP | 22.1 (1.5) | 22.8 (2.2) | 1.6 | 0.2895 |
| SAA | 21.7 (1.2) | 22.8 (2.4) | 2.1 | 0.0794 |
| IL-6 | 0.07 (1.1) | 0.98 (1.7) | 1.9 | 0.0314 * |
| IL-10 | -2.3 (1.1) | -1.7 (1.0) | 1.5 | 0.0272 * |
| IL-17A | -2.1 (1.3) | -0.3 (1.5) | 3.3 | <0.0001 * |
| IL-17F | 0.05 (1.1) | 1.3 (1.5) | 2.4 | 0.0007 * |
| IL-22 | 1.9 (1.1) | 3.1 (1.3) | 2.4 | 0.0002 * |
| TNFα | 0.5 (0.75) | 1.4 (1.1) | 1.8 | 0.0002 * |
| IFNɣ | 2.4 (0.84) | 2.9 (1.3) | 1.5 | 0.0259 * |
Data are mean (SD); *p<0.05 and |fold difference| >1.4; ▫adjusted for confounding factors age & sex.
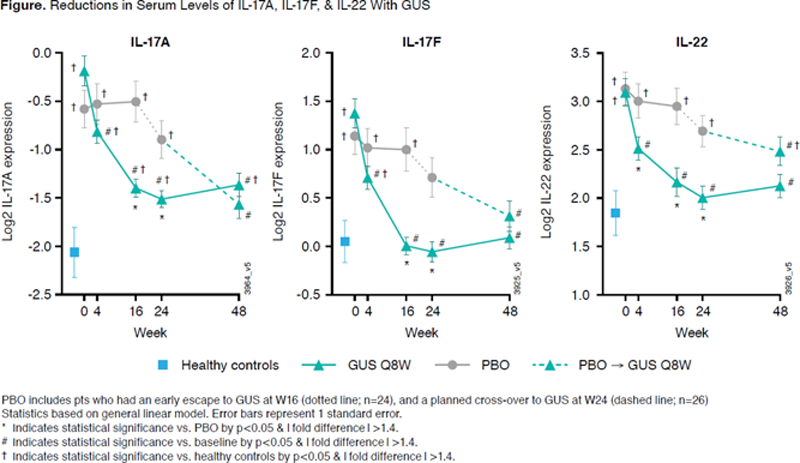
Conclusion: GUS-tx TNFi-IR pts showed response-specific associations with BL biomarkers (IL-22, IFNɣ, IL-6, and SAA). GUS resulted in decreased levels of elevated CRP, SAA, IL-6, IL-17A, IL-17F, and IL-22, while no significant change was observed with PBO tx. Reductions in these biomarkers were evident as early as W4 and approximated levels seen in HC from W16 onward (W4 for IL-22), suggesting apparent normalization of effector cytokines associated with the IL-23/Th17 axis following GUS tx.
REFERENCES:
[1]Deodhar A et al. Lancet 2020;395:1115-25.
[2]Mease PJ et al. Lancet 2020;395:1126-36.
[3]Coates LC et al. doi:10.1136/annrheumdis-2021-220991.
Disclosure of Interests: Georg Schett Speakers bureau: Amgen, AbbVie, Bristol Myers Squibb, Eli Lilly, Gilead, Janssen, Novartis and UCB, Warner Chen Shareholder of: Janssen, Employee of: Janssen, Sheng Gao Shareholder of: Janssen, Employee of: Janssen, Soumya D Chakravarty Shareholder of: Janssen, Employee of: Janssen, May Shawi Shareholder of: Janssen, Employee of: Janssen, Frederic Lavie Shareholder of: Janssen, Employee of: Janssen, Elke Theander Shareholder of: Janssen, Employee of: Janssen, Marlies Neuhold Shareholder of: Janssen, Employee of: Janssen, Laura Coates Speakers bureau: AbbVie, Amgen, Biogen, Celgene, Eli Lilly, Galapagos, Gilead, Janssen, Medac, Novartis, Pfizer and UCB, Consultant of: AbbVie, Amgen, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Eli Lilly, Gilead, Galapagos, Janssen, Novartis, Pfizer, and UCB, Grant/research support from: AbbVie, Amgen, Celgene, Eli Lilly, Janssen, Novartis, Pfizer, and UCB, Stefan Siebert Speakers bureau: AbbVie, Biogen, GSK, Janssen, Novartis, UCB, Grant/research support from: AbbVie, Amgen, Boehringer Ingelheim, Bristol Myers Squibb, GSK, Janssen, Novartis, UCB