

Background: The most frequent form of vasculitis in elderly people is giant cell arteritis (GCA) with an annual incidence rate less than 10 per 100,000 persons over the age of 50. Like most vasculitides, GCA is characterized by chronicity and relapses, leading to significant overall morbidity and higher mortality in a subset of patients with aortic involvement and dissection. Most studies carried out so far have been retrospective, used monocentric study designs and small patient cohorts. Therefore, the Joint Vasculitis Registry in German-speaking countries (GeVas) has been established to record patients, who have been recently diagnosed with vasculitis or who have changed their treatment due to a relapse (inception cohort). The GeVas-Registry allows a long-term follow-up of a substantial cohort of vasculitis patients in a prospective and multicenter manner.
Objectives: To describe the subgroup of GCA and its characteristics within the GeVas registry.
Methods: GeVas is a prospective, web-based, multicenter, clinician-driven registry for the documentation of organ manifestations, damage, long-term outcomes, and therapy regimens in various types of vasculitis. Recruitment started in June 2019. By January 2022, 17 centers in Germany were initiated and have begun enrolling patients. Meanwhile, more than 350 patients have been documented in the registry. Sites in Austria and the German-speaking cantons of Switzerland will be integrated soon (1).
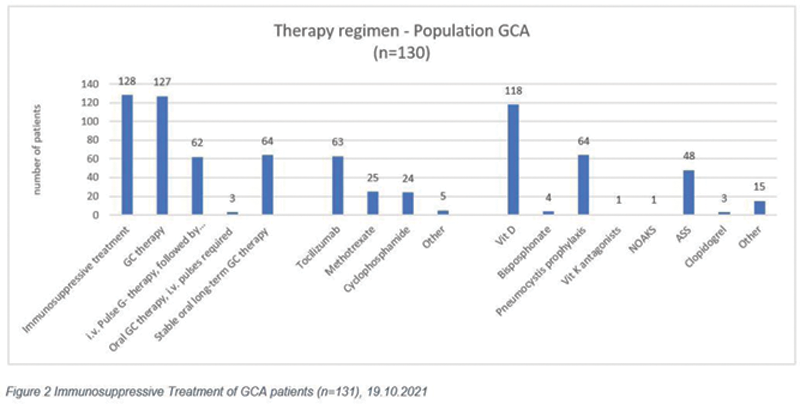
Results: By mid-October 2021, the participating centers recruited 131 GCA patients into the registry. 21.7% of patients (n=28) were enrolled in the registry due to relapse, and 78.3% (n=101) due to a first-time diagnosis. In accordance with long-standing epidemiology data, the majority of patients (67,2%), were female (n=88), and 32.8% (n=43) were male. Mean age was 74 years (max. 92y, min. 52y). The most frequently recorded organ manifestations in GCA patients addressed cranial and ophthalmic symptoms, and the cardiovascular system. However, vascular lung/chest involvement was also observed in 3% of cases (n=4). Out of the 131 patients, 97.7% (n=128) received immunosuppressive therapy, three refused to take any medication. An equal number of patients were treated with glucocorticoid (GC) therapy. While about two equal parts were treated by stable long term oral GC therapy (47,7%, n=62) or by i.v. pulse therapy followed by tapering (49,2%, n=64), only about 2.3% (n=3) were treated by oral GC therapy with intermittent i.v. pulses. 48.5% (n=63) of patients received tocilizumab as additional immunosuppressive therapy, 19.2% (n=25) methotrexate, and 18.5% (n=24) cyclophosphamide i.v. pulses.
Conclusion: In June 2019, we successfully established the prospective multicenter vasculitis registry GeVAS. It describes the first systematically recorded prospective GCA cohort in German-speaking countries. Its characteristics correspond to those that can be expected from the literature, with some unexpected finding e.g. the high proportion of patients treated with cyclosphosphamid, probably reflecting a sicker patient population with e.g. aortic or central nervous involvement. After 2.5 years of follow-up documentation, the first long-term results will be systematically evaluated and interpreted. The newly acquired data on disease manifestation, diagnostics and therapy regimens will provide important insights into the treatment of GCA patients in Germany and may generate further research goals.
REFERENCES:
Trial registration: German Clinical Trials Register (Deutsches Register Klinischer Studien): DRKS00011866. Registered 10 May 2019. 3
[1]C Iking-Konert; P Wallmeier; S Arnold; S Adler; K de Groot; B Hellmich; B Hoyer; K Holl-Ulrich; Ihorst; M Kaufmann; I Kötter; U Müller-Ladner; T Magnus; J. Rech; H. Schulze-Koops; N. Venhoff; T. Wiech; P. Villiger; F. Schubach; P. Lamprecht. The Joint Vasculitis Registry in German-speaking countries (GeVas) – a prospective, multicenter registry for the follow-up of long-term outcomes in vasculitis. BMC Rheumatol. 2021 Jul 31;5(1):40. doi: 10.1186/s41927-021-00206-2.
Acknowledgements: GeVas was supported by unrestricted grants by: DGRh, John Grube Foundation, Vifor and Roche Pharma
Disclosure of Interests: Pia Wallmeier: None declared, Sabrina Arnold: None declared, Fabian Schubach: None declared, Gabriele Ihorst: None declared, Peer Aries: None declared, Raoul Bergner Consultant of: Advisory Board VIFOR, Grant/research support from: John-Grube Research Award 2021, Jan Philip Bremer: None declared, Norman Görl: None declared, Bernhard Hellmich: None declared, Jörg Henes: None declared, Bimba Hoyer: None declared, Antje Kangowski: None declared, Ina Kötter: None declared, Tim Magnus: None declared, Claudia Metzler: None declared, Ulf Müller-Ladner: None declared, Matthias Schaier: None declared, Ulf Schönermark: None declared, Jens Thiel: None declared, Leonore Unger: None declared, Nils Venhoff Speakers bureau: Roche and Vifor, Consultant of: Roche and Vifor, Grant/research support from: John-Grube Research Award 2021, Julia Weinmann-Menke: None declared, Jana Petersen: None declared, Peter Lamprecht Speakers bureau: Lecture fees from: Chugai, GSK, Roche, Consultant of: Consulting & lecture fees from: Chugai, GSK, Roche, and Vifor., Grant/research support from: Research grants for GeVas: DGRh, John Grube Foundation, Roche, and Vifor, Christof Iking-Konert Speakers bureau: lecture fees from: Chugai, GSK, Roche, and Vifor., Consultant of: Consulting fees from: Chugai, GSK, Roche, and Vifor., Grant/research support from: Research grants for GeVas: DGRh, John Grube Foundation, Roche, and Vifor;