

Background: Ixekizumab (IXE), a monoclonal antibody that selectively targets interleukin IL-17A, has shown efficacy in patients with radiographic axial spondyloarthritis (r-axSpA). Spinal pain, in particular spinal pain at night (SP-N), is a major contributor to the patient burden of r-axSpA.
Objectives: To assess SP-N improvement in patients up to week (W) 52 and to determine the association of SP-N improvement in patients treated with IXE with other patient-reported outcomes (PROs) at W16 and with reaching ASDAS LDA at W52.
Methods: The Phase III COAST-V (NCT02696785) trial investigated the efficacy of IXE in 341 patients with r-axSpA and were biological disease-modifying anti-rheumatic drug (bDMARD)-naïve. Patients were randomised to IXE every 2W (IXEQ2W), IXE every 4W (IXEQ4W), adalimumab (ADA) or placebo (PBO) up to W16. Only approved dose IXEQ4W data are presented here. SP-N was measured at each visit using a numeric rating scale (NRS) (0-10). A clinically relevant improvement in SP-N was defined as >2 point improvement from baseline. Differences in baseline variables between those achieving versus not achieving >2 improvement in SP-N were tested using Fisher’s exact test (binary variables) and analysis of variance (ANOVA; continuous variables). Associations of SP-N improvement with PROs (BASFI, Fatigue Severity NRS, Jenkins Sleep Evaluation Questionnaire (JSEQ), SF-36 PCS) at W16, and ASDAS LDA at W52 were tested using analysis of covariance (ANCOVA; continuous variables) and logistic regression (binary variables). Missing values were imputed using non-responder imputation, and modified baseline observation carried forward.
Results: A greater proportion of patients achieved >2 improvement in SP-N with IXE treatment compared to PBO at W16 (63.0% vs. 32.2%, p <0.001) and improvement was sustained up to W52 (
Baseline demographics, clinical characteristics, and PROs of IXE treated patients achieving vs. not achieving >2 improvement in SP-N at W16.
| Achieved >2 Improvement in SP-N at W16 | Yes (n=51 ) | No (n=30 ) | ||
|---|---|---|---|---|
| Baseline Characteristics | ||||
| Age, yrs | 38.6 (11.4)* | 44.9 (12.4) | ||
| Positive for HLA-B27, n (%) | 50.0 (98.0)* | 25.0 (83.3) | ||
| CRP (mg/L) | 14.9 (14.9)* | 7.6 (8.4) | ||
| ASDAS | 3.9 (0.8)* | 3.5 (0.6) | ||
| Spinal Pain at Night | 7.4 (1.3)* | 6.4 (1.4) | ||
| PROs | Baseline | W16 CFB | Baseline | W16 CFB |
| BASFI | 6.1 (1.9) | -3.4 (2.2)*** | 6.0 (1.6) | -0.7 (1.5) |
| Fatigue Severity NRS | 6.9 (1.7) | -3.5 (2.6)*** | 6.3 (1.6) | -0.7 (2.0) |
| JSEQ | 7.1 (5.4) | -3.2 (4.2)*** | 7.2 (5.2) | -0.1 (2.7) |
| SF-36 PCS | 32.8 (7.7) | 10.9 (7.7)*** | 36.1 (6.7) | 2.0 (6.5) |
| ASDAS <2.1 at W52 Response, n (%) | 34 (66.7)** | 9 (33.3) | ||
Values represent mean (SD) unless otherwise stated. *p<0.05, **p<0.01, ***p<0.001 versus those not achieving >2 improvement in SP-N.
HLA-B27; Human Leukocyte antigen-B27, CRP; C-reactive protein, ASDAS; Ankylosing Spondylitis Disease Activity Score, CFB; change from baseline, BASFI; Bath Ankylosing Spondylitis Functional Index, NRS; Numeric Rating Score, JSEQ; Jenkins Sleep Evaluation Questionnaire, SF-36 PCS; Short-Form 36 physical component score.
Patients achieving meaningful spinal pain at night improvement through W52.
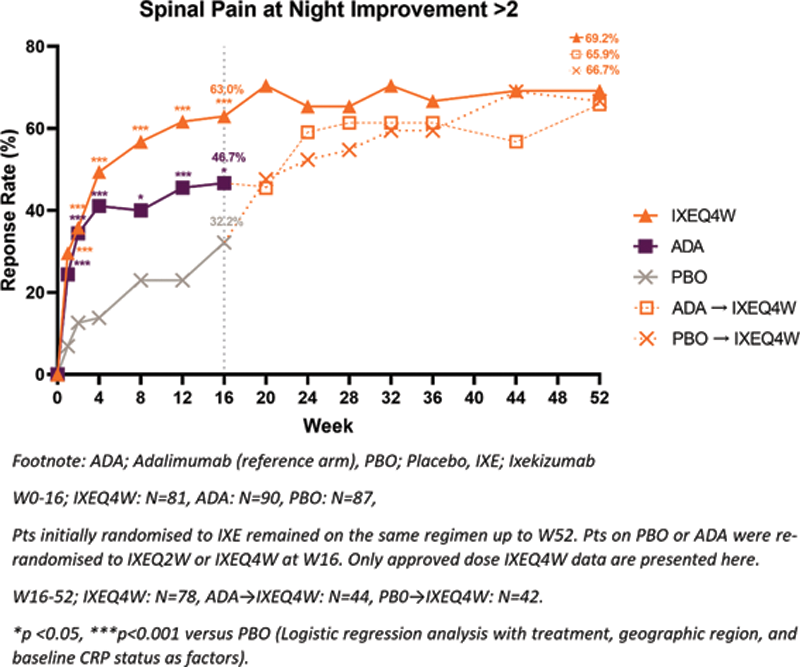
Conclusion: IXE improved SP-N for patients with r-axSpA not previously treated with bDMARDs. Improvements in SP-N were associated with improvements in disease activity, function, fatigue and quality of life.
Disclosure of Interests: Sofia Ramiro Speakers bureau: Eli Lilly and Company, MSD, Novartis, UCB, Consultant of: AbbVie, Eli Lilly and Company, MSD, Novartis, Pfizer, UCB, Sanofi, Grant/research support from: AbbVie, Galapagos, Pfizer, Novartis, UCB, Cédric Lukas Speakers bureau: AbbVie, Amgen, Novartis, Pfizer, Galapagos, Consultant of: AbbVie, BMS, Chugai, Roche, Eli Lilly and Cmpany, MSD, Novartis, Pfizer, Amgen, Grant/research support from: Roche, Pfizer, Novartis, Eli Lilly and Company, Michael J. Nissen Speakers bureau: Eli Lilly and Company, Janssen, Novartis, Consultant of: AbbVie, Celgene, Eli Lilly and Company, Janssen, Novartis, Pfizer, Yves Schymura Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Khai Ng Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Andrew Bradley Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Gabriel Doridot Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Soyi Liu Leage Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Antoni Chan Speakers bureau: Novartis, AbbVie, Amgen, UCB, Janssen, Celgene, Biogen, Consultant of: Novartis, Eli Lilly and Company, Janssen, Amgen, UCB, AbbVie, James Cheng-Chung Wei Consultant of: tsh biopharma, Abbvie, BMS, Celgene, Chugai, Eisai, Janssen, Novartis, Pfizer, Sanofi-Aventis, UCB pharma, Grant/research support from: AbbVie, BMS, Celgene, Eli Lilly and Company, Janssen, Novartis, Pfizer, UCB pharma