

Background: The treatment landscape in psoriatic arthritis (PsA) is changing, including newer biologic and targeted synthetic disease-modifying antirheumatic drugs (b/tsDMARDs) with different modes of action becoming available. However, the most effective treatment strategy in routine care remains to be established.
Objectives: To explore the uptake and treatment patterns of newer b/tsDMARDs, namely JAK-inhibitors (JAKi; baricitinib, tofacitinib, upadacitinib), IL-17-inhibitors (ixekizumab, secukinumab), abatacept, apremilast, and ustekinumab in PsA patients from the Nordic countries. Furthermore, to describe patient characteristics and extra-musculoskeletal manifestations at treatment start (=baseline).
Methods: Observational cohort study, using prospectively collected routine care data from 4 Nordic rheumatology registries. Treatments (newer b/tsDMARDs with tumor-necrosis-factor inhibitors (TNFi) as the reference) initiated from January 2009 until December 2020 and corresponding baseline patient characteristics were identified. Linkage to national patient registries was used to identify previous extra-musculoskeletal manifestations (0-5 years). Country-level data were pooled for analyses. Uptake of each drug was explored as the cumulative number of treatment starts (a) overall, irrespective of previous b/tsDMARD experience, and (b) in b/tsDMARD-naïve patients. Each patient could contribute >1 treatment course.
Results: Overall, 13,364 unique patients contributing 24,325 treatment courses with either a newer b/tsDMARD (4,855, 20%) or a TNFi (19,470, 80%, whereof 10,897 were started year 2015-20) were identified. For the sub-group of 11,892 first b/tsDMARD treatment courses, 1,009 (8%) were a newer b/tsDMARD (10,883 were a TNFi, whereof 5,956 were started year 2015-20).
Secukinumab dominated the newer b/tsDMARD uptake (1,848 new-starts,
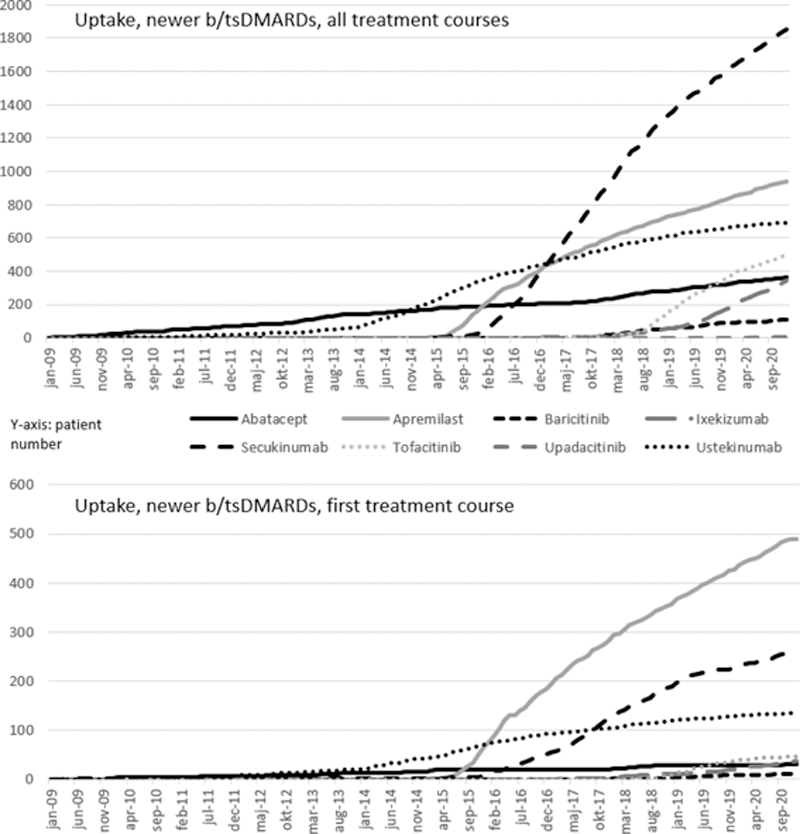
Patients starting a newer b/tsDMARD tended to have longer disease duration and slightly higher disease activity at baseline (DAS28, patient-reported outcomes) than TNFi initiators (
Baseline characteristics upon treatment start
| Abata-cept | Apre-milast | Bari-citinib | Ixe-kizumab | Secuki-numab | Tofa-citinib | Upada-citinib | Uste-kinumab | Any TNFi | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Cumulative uptake, n | 362 | 935 | 106 | 342 | 1848 | 494 | 6 | 691 | 19470 | |
| Male gender, % | 33 | 42 | 27 | 38 | 40 | 33 | 33 | 37 | 44 | |
| Age | 54 (12) | 53 (12) | 55 (13) | 52 (13) | 51 (13) | 54 (13) | 52 (10) | 50 (12) | 49 (13) | |
| b/tsDMARD treatment number, % | 1 | 9 | 52 | 9 | 11 | 14 | 9 | 0 | 20 | 56 |
| 2 | 19 | 15 | 12 | 26 | 25 | 18 | 17 | 19 | 25 | |
| ≥3 | 72 | 33 | 78 | 74 | 61 | 73 | 83 | 62 | 19 | |
| Disease duration, yrs | 9 (8) | 8 (8) | 10 (8) | 10 (8) | 9 (9) | 11 (10) | 8 (8) | 8 (9) | 7 (8) | |
| Pain, VAS (0-100) | 63 (21) | 61 (23) | 64 (23) | 64 (25) | 63 (24) | 66 (23) | 75 (17) | 64 (23) | 59 (24) | |
| DAS28 | 4.73 (1.34) | 4.04 (1.35) | 3.95 (1.36) | 4.24 (1.19) | 4.13 (1.36) | 4.49 (1.33) | 4.74 (0.88) | 4.19 (1.32) | 4.07 (1.29) | |
| Uveitis, %* | 3 | 2 | 3 | 1 | 2 | 3 | 0 | 2 | 2 | |
| IBD, %* | 1 | 1 | 3 | 1 | 1 | 1 | - | 3 | 1 | |
Numbers are mean (SD) unless otherwise stated
IBD: inflammatory bowel disease, bDMARD: biologic DMARD, ts: targeted synthetic
*0-5 years previously, available all study period for Iceland, Sweden, Finland until 31Dec2018, not available for Denmark
Conclusion: In this cross-country collaboration we were able to explore uptake of newer b/tsDMARDs. TNFi still dominates compared to newer b/tsDMARDs in routine care treatment of PsA. Newer b/tsDMARDs are mainly used in patients with several previous treatment failures, i.e. with longer disease duration and higher disease activity, indicating difficult to treat disease. Further studies are planned to explore real-world treatment patterns and outcomes.
Acknowledgements: BG and DdiG contributed equally.
Partly funded by NordForsk and Foreum grants. On behalf of the Danish DANBIO, Swedish SRQ, Norwegian NOR-DMARD, Finnish ROB-FIN and Icelandic ICEBIO registries
Disclosure of Interests: Bente Glintborg Grant/research support from: Pfizer, AbbVie, BMS, Daniela Di Giuseppe: None declared, Johan K Wallman Consultant of: AbbVie, Amgen, Celgene, Eli Lilly, Novartis, Grant/research support from: AbbVie, Amgen, Eli Lilly, Novartis, Pfizer, Dan Nordström: None declared, Björn Gudbjornsson Speakers bureau: Novartis and Amgen, Merete Lund Hetland Grant/research support from: AbbVie, Biogen, BMS, Celltrion, Eli Lilly Denmark A/S, Janssen Biologics B.V, Lundbeck Fonden, MSD, Pfizer, Roche, Samsung Biopis, Sandoz, Novartis., Johan Askling Grant/research support from: Abbvie, Astra-Zeneca, BMS, Eli Lilly, MSD, Pfizer, Roche, Samsung Bioepis, Sanofi, and UCB., Gerdur Gröndal: None declared, Tuulikki Sokka-Isler Grant/research support from: Abbvie, Amgen, BMS, Celgene, Eli Lilly, GSK, Medac, MSD, Novartis, Orion Pharma, Pfizer, Roche, Sandoz, UCB, Sella Aarrestad Provan: None declared, Ulf Lindström: None declared