

Background: Risankizumab (RZB) is a monoclonal antibody that specifically inhibits interleukin 23.
Objectives: To evaluate the achievement of Minimal Disease Activity (MDA), its components, and achievement of Disease Activity in PsA Low Disease Activity and Remission (DAPSA LDA+REM, [DAPSA score ≤14]) in patients receiving RZB or placebo (PBO) in the KEEPsAKE 1 and 2 clinical trials.
Methods: KEEPsAKE-1 and -2, double-blind, phase 3 trials, evaluated the efficacy of RZB versus PBO for the treatment of adult patients with active psoriatic arthritis (PsA). Patients were randomized (1:1) to receive subcutaneous RZB 150 mg or PBO at weeks 0, 4, and 16. The open label extension began at Week 24 with all patients receiving RZB 150 mg every 12 weeks thereafter. Achievement of MDA, its components, and achievement of DAPSA LDA+REM are reported using non-responder imputation.
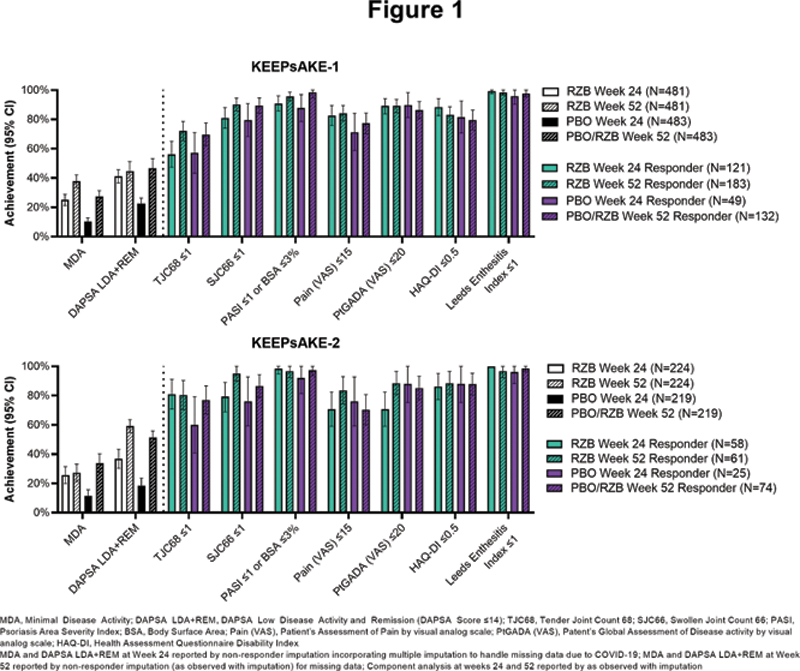
Results: MDA achievement at Week 52 in KEEPsAKE-1 was 37.9% for patients originally randomized to RZB and 27.4% for patients originally randomized to PBO. In KEEPsAKE-2, MDA achievement was 27.2% and 33.8% for patients originally randomized to RZB and PBO, respectively. Achievement of MDA and its components are presented in
Conclusion: Patients treated with RZB demonstrate achievement of MDA, its components, and DAPSA LDA+REM at Weeks 24 and 52.
Acknowledgements: AbbVie Inc, participated in the study design; study research; collection, analysis and interpretation of data; and writing, reviewing, and approving of this abstract for submission. AbbVie funded the research for this study and provided writing support for this abstract. Medical writing assistance was provided by Trisha Rettig, Ph.D. of AbbVie
Disclosure of Interests: Joseph F. Merola Consultant of: Amgen, Bristol-Myers Squibb, Abbvie, Dermavant, Eli Lilly, Novartis, Janssen, UCB, Sanofi, Regeneron, Sun Pharma, Biogen, Pfizer and Leo Pharma, Iain McInnes Consultant of: AbbVie, Amgen, Astra Zeneca, Compugen, Cabaletta, Evelo, Janssen, Lilly, Novartis, Pfizer, Sanofi, and UCB, Grant/research support from: AbbVie, Amgen, Astra Zeneca, Janssen, Lilly, Novartis, Pfizer, UCB, Arthur Kavanaugh Consultant of: AbbVie Inc., Amgen, Astra-Zeneca, BMS, Celgene, Centocor-Janssen, Pfizer, Roche, and UCB, Grant/research support from: AbbVie Inc., Amgen, Astra-Zeneca, BMS, Celgene, Centocor-Janssen, Pfizer, Roche, and UCB, Peter Nash Speakers bureau: Abbvie, Amgen, Janssen, Lilly, Novartis, Pfizer, UCB, BMS, Rocje, Sanofi, Gilead/Galapagos, MSD, Samsung, Celgene, Amgen, Boehringer, Consultant of: Abbvie, Amgen, Janssen, Lilly, Novartis, Pfizer, UCB, BMS, Rocje, Sanofi, Gilead/Galapagos, MSD, Samsung, Celgene, Amgen, Boehringer, Grant/research support from: Abbvie, Amgen, Janssen, Lilly, Novartis, Pfizer, UCB, BMS, Rocje, Sanofi, Gilead/Galapagos, MSD, Samsung, Celgene, Amgen, Boehringer, Zhenyi Xue Shareholder of: AbbVie Inc., Employee of: AbbVie Inc., Vassilis Stakias Shareholder of: AbbVie Inc., Employee of: AbbVie Inc., Ann Eldred Shareholder of: AbbVie Inc., Employee of: AbbVie Inc., Sandra Ciecinski Shareholder of: AbbVie Inc., Employee of: AbbVie Inc., Kevin Douglas Shareholder of: AbbVie Inc., Employee of: AbbVie Inc., Laura Coates Speakers bureau: AbbVie, Amgen, Biogen, Celgene, Eli Lilly, Galapagos, Gilead, Janssen, Medac, Novartis, Pfizer and UCB, Consultant of: AbbVie, Amgen, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Eli Lilly, Gilead, Galapagos, Janssen, Novartis, Pfizer and UCB, Grant/research support from: AbbVie, Amgen, Celgene, Eli Lilly, Janssen, Novartis and Pfizer