

Background: Guselkumab (GUS), an anti-IL23p19-subunit mAb, demonstrated significant efficacy vs placebo (PBO) in achieving ACR20 response at week (W) 24 in patients (pts) with active PsA in phase 3 trials, DISCOVER-1&2 (D1&2). 1,2 Pts with PsA report pain relief as a treatment priority. 3
Objectives: To assess GUS treatment effect through 2 years on pt-reported pain across outcome measures.
Methods: Pts with active PsA in D1 (n=381; 31% received 1-2 prior TNFi) and D2 (n=739; biologic-naïve) were randomized (1:1:1) to GUS 100 mg every 4 weeks (Q4W); GUS 100 mg at W0, W4, then Q8W; or PBO with crossover to GUS 100 mg Q4W at W24 (PBO→Q4W). Pts rated pain using visual analog scale (VAS; 0-10; Pt Pain) and reported Bodily Pain intensity over past 4 W via 36-Item Short-Form Health Survey question 21 (0-5). Pts with spondylitis and peripheral arthritis at baseline (BL) rated Spinal and Joint Pain (0-10). Observed mean change, % improvement from BL in tender (TJC; 0-68) and swollen joint counts (SJC; 0-66), and proportion of pts achieving ≥20/≥50% improvement in Pt Pain (nonresponder imputation [NRI]) were evaluated.
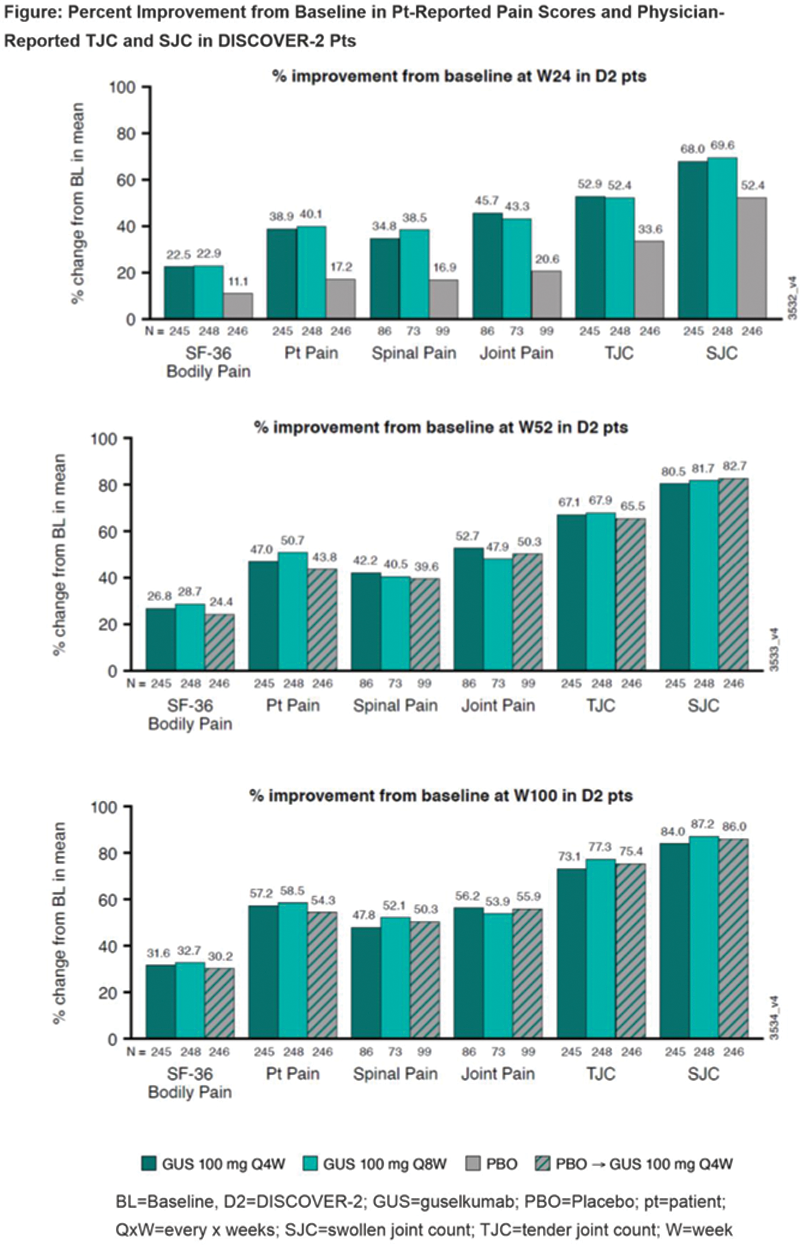
Results: In D2, mean BL for Bodily Pain (range: 4.4-4.5), Pt Pain (6.2-6.3), Spinal Pain (6.5-6.7), Joint Pain (6.3-6.8), SJC (11.7-12.9) and TJC (19.8-22.4) indicated moderate pain and disease activity at study outset. GUS-treated pts reported ~2x improvement in Pt Pain, Spinal Pain, Joint Pain, and Bodily Pain intensity at W24 vs PBO, which were maintained or increased at W52 and W100. PBO→Q4W pts had similar improvements. Pt-reported pain appeared more sensitive to treatment effect, with larger differences in % improvement vs PBO than physician-reported TJC/SJC at W24. D1 showed consistent results through 1 year. In 748 GUS-treated pts across D1&2, substantial proportions achieved meaningful improvement in Pt Pain at early time points: 32% (W4) and 48% (W8) achieved ≥20% improvement; 28% (W12) and 33% (W16) achieved ≥50% improvement. At W24, 63%/39% reported ≥20%/≥50% improvement in pain.
Conclusion: GUS provided consistent and durable improvements in pt-reported pain across several outcome measures. Pt-reported pain as an early and sensitive indicator of treatment effect in pts with active PsA and additional factors merits further evaluation.
REFERENCES:
[1]Deodhar A et al. Lancet. 2020;395:1115-25
[2]Mease P et al. Lancet . 2020;395:1126-36
[3]Gudu T et al. Expert Rev Clin Immunol . 2018;14:405-17
Observed Mean (SD) Change from Baseline in Pain Scores, TJC, and SJC at W24, W52, and W100 in DISCOVER-2
| W24 | W52 | W100 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| GUS Q4W | GUS Q8W | PBO | GUS Q4W | GUS Q8W | PBO→Q4W | GUS Q4W | GUS Q8W | PBO→Q4W | |
| Pt Pain (0-10) ,* N | 240 | 243 | 243 | 229 | 234 | 231 | 220 | 224 | 215 |
| -2.39 (2.35) | -2.53 (2.47) | -1.08 (2.42) | -2.89 (2.68) | -3.20 (2.56) | -2.75 (2.66) | -3.52 (2.62) | -3.69 (2.63) | -3.41 (2.58) | |
| Spinal Pain (0-10), + N | 80 | 65 | 92 | 79 | 64 | 88 | 76 | 61 | 82 |
| -2.26 (2.57) | -2.54 (2.70) | -1.13 (2.48) | -2.74 (2.63) | -2.67 (2.71) | -2.65 (2.69) | -3.11 (2.67) | -3.44 (2.71) | -3.37 (2.66) | |
| Joint Pain (0-10), + N | 80 | 65 | 92 | 79 | 64 | 88 | 76 | 61 | 82 |
| -2.88 (2.17) | -2.90 (2.68) | -1.40 (2.91) | -3.32 (2.27) | -3.21 (2.76) | -3.42 (2.92) | -3.54 (2.35) | -3.61 (2.77) | -3.80 (2.95) | |
| SF-36 (Q21; 0-5) , N | 240 | 243 | 242 | 229 | 234 | 230 | 220 | 224 | 214 |
| -0.99 (1.03) | -1.03 (1.12) | -0.50 (1.11) | -1.18 (1.33) | -1.29 (1.17) | -1.10 (1.16) | -1.39 (1.25) | -1.47 (1.38) | -1.36 (1.27) | |
| TJC (0-68) , N | 240 | 243 | 243 | 228 | 234 | 231 | 220 | 224 | 213 |
| -11.85(9.88) | -10.37(9.49) | -7.26(11.15) | -15.04(10.51) | -13.44(10.03) | -14.15(11.39) | -16.37(10.70) | -15.27(11.10) | -16.29(11.27) | |
| SJC (0-66) , N | 240 | 243 | 243 | 228 | 234 | 231 | 220 | 224 | 213 |
| -8.77(5.46) | -8.14(6.07) | -6.44(7.20) | -10.38(6.17) | -9.56(6.28) | -10.17(6.79) | -10.83(6.66) | -10.20(6.88) | -10.58(6.15) | |
*ACR, DAPSA, MDA: VAS 0-10; + BASDAI: VAS 0-10
ACR=American College of Rheumatology; BASDAI=Bath Ankylosing Spondylitis Disease Activity Index; DAPSA=Disease Activity in Psoriatic Arthritis; GUS=guselkumab; MDA=minimal disease activity; PBO=placebo; pt=patient; Q=question; QxW=every x week; SD=standard deviation; SF-36=36-Item Short-Form Survey; SJC=swollen joint count; TJC=tender joint count; VAS=visual analog scale; W=week
Disclosure of Interests: Peter Nash Consultant of: AbbVie, Bristol Myers Squibb, Boehringer, Celgene, Eli Lilly, Gilead, Janssen, Novartis, Pfizer, Roche, Sandoz, and Sun Pharma, Grant/research support from: AbbVie, Bristol Myers Squibb, Boehringer, Celgene, Eli Lilly, Gilead, Janssen, Novartis, Pfizer, Roche, Sandoz, and Sun Pharma, Lai-Shan Tam Consultant of: AbbVie, Boehringer Ingelheim, Eli Lilly, Janssen, Pfizer, and Sanofi, Grant/research support from: Amgen, Boehringer Ingelheim, GSK, Janssen, Novartis, and Pfizer, Wen-Chan Tsai Consultant of: AbbVie, Eli Lilly, Novartis, Pfizer, and Janssen, Ying Ying Leung Consultant of: AbbVie, Eli Lilly, Janssen, and Novartis and on advisory board for Janssen, Daniel Furtner Shareholder of: Johnson & Johnson, Employee of: Janssen, a division of Johnson & Johnson Pte. Ltd, Shihong Sheng Shareholder of: Johnson & Johnson, Employee of: Janssen Research and Development, LLC, Yanli Wang Consultant of: Janssen, Employee of: Cytel, Inc., May Shawi Shareholder of: Johnson & Johnson, Employee of: Janssen Global Services, LLC, Alexa Kollmeier Shareholder of: Johnson & Johnson, Employee of: Janssen Research and Development, LLC, Jonathan Sherlock Shareholder of: Johnson & Johnson, Employee of: Janssen Research and Development, LLC, Daniel Cua Shareholder of: Johnson & Johnson, Employee of: Janssen Research and Development, LLC