

Background: Ixekizumab (IXE), an IL-17A inhibitor, has demonstrated efficacy in clinical trials 1-3 but real-world effectiveness (RWE) data are limited. 4
Objectives: To describe changes in disease activity and patient-reported outcomes (PROs) at 6 and 12 months follow-up among psoriatic arthritis (PsA) patients initiating IXE in a routine clinical setting.
Methods: This retrospective cohort study included patients from the OM1 PsA Registry (OM1, Boston, MA), a linked electronic medical record and administrative claims dataset with over 50,000 patients. Eligible patients had ≥1 prescription for IXE (first = index), were ≥18 years old at index, had ≥1 diagnosis code for PsA in the 12 months before or on index, and had ≥12 months of baseline and ≥6 months of follow-up data as of June 2021. For patients with baseline and follow-up measures available, changes in Clinical Disease Activity Index (CDAI), PROs, and other clinical outcomes from baseline to 6 and 12 months were described. For patients on IXE monotherapy, change in CDAI score from baseline to 6 and 12 months was assessed using mixed effects linear models adjusted for age, sex, and baseline CDAI score.
Results: The study population included 1,812 patients with a mean age of 53.7 years (
Demographic and Clinical Characteristics by Therapy Status
| All Patients(N=1,812 ) | Monotherapy(N=1,485 ) | Combination Therapy(N=327 ) | ||
|---|---|---|---|---|
| Age (years) | Mean (s.d.) | 53.7 (12.2) | 53.9 (12.3) | 52.9 (11.7) |
| Median (Q1-Q3) | 55 (46-62) | 55 (46-62) | 54 (45-61) | |
| Sex | Female | 1,108 (61.1%) | 909 (61.2%) | 199 (60.9%) |
| Male | 704 (38.9%) | 576 (38.8%) | 128 (39.1%) | |
| Charlson Comorbidity Index | Mean (s.d.) | 1.3 (1.6) | 1.3 (1.6) | 1.5 (1.7) |
| Median (Q1-Q3) | 1 (0-2) | 1 (0-2) | 1 (0-2) | |
| BMI | Underweight: <18.5 | 10 (0.6%) | 10 (0.7%) | 0 (0.0%) |
| Normal weight: 18.5-24.9 | 210 (12.2%) | 172 (12.2%) | 38 (12.1%) | |
| Overweight: 25-29.9 | 455 (26.5%) | 379 (27.0%) | 76 (24.2%) | |
| Obese: >= 30 | 1,045 (60.8%) | 845 (60.1%) | 200 (63.7%) | |
| Missing | 92 | 79 | 13 | |
| Domains of PsA: Psoriasis | Yes | 1,490 (82.2%) | 1,222 (82.3%) | 268 (82.0%) |
| No | 322 (17.8%) | 263 (17.7%) | 59 (18.0%) | |
| Domains of PsA: Enthesitis | Yes | 510 (28.1%) | 409 (27.5%) | 101 (30.9%) |
| No | 1,302 (71.9%) | 1,076 (72.5%) | 226 (69.1%) | |
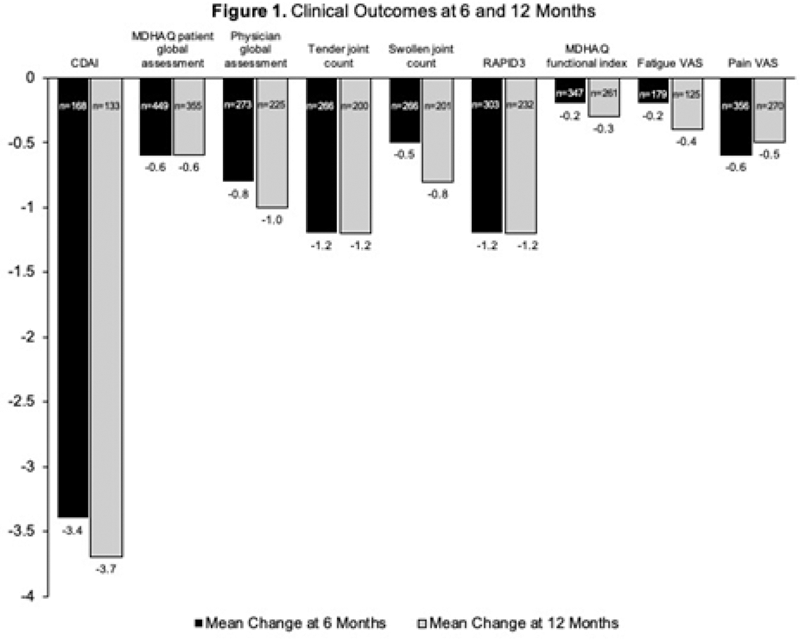
Of patients with a baseline CDAI score, 61% had moderate or severe disease activity. For all patients, CDAI scores improved (decreased) by an average of 3.4 and 3.7 points at 6 and 12 months, respectively, from a baseline mean of 15.4. All disease activity measures and PROs improved from baseline to 6 and 12 months (
Conclusion: In this cohort of PsA patients with multiple prior b/tsDMARD failures, improvements in disease activity and PROs were observed at 6 and 12 months after initiating treatment with IXE. Improvements were observed in patients overall and in the monotherapy subgroup. More real-world research on IXE and other bDMARDs are important to understand the effect of treatment choices on clinical and PROs in both bDMARD-naive and experienced PsA patients.
REFERENCES:
[1]Mease PJ. Ann. Rheum. Dis. 2017;76(1):79-87
[2]Nash P. Lancet . 2017;389(10086):2317-2327
[3]Mease PJ. Ann. Rheum. Dis. 2020;79(1):123-131
[4]Berman J. Biologics . 2021 Nov 18;15:463-470
Disclosure of Interests: Julie Birt Shareholder of: Shareholder of Eli Lilly and Company, Employee of: Employee of Eli Lilly and Company, William Tillett Speakers bureau: Abbvie, Amgen, Celgene, Eli-Lilly, Janssen, MSD, Novartis,, Pfizer, UCB, Consultant of: Abbvie, Amgen, Celgene, Eli-Lilly, Janssen, MSD, Novartis, Pfizer, UCB, Grant/research support from: Abbvie, Amgen, Celgene, Eli-Lilly, Janssen, UCB, Cristi Cavanaugh: None declared, Yoojin Jung: None declared, Aisha Vadhariya Shareholder of: Shareholder of Eli Lilly and Company, Employee of: Employee of Eli Lilly and Company, Sarah Ross Shareholder of: Shareholder of Eli Lilly and Company, Employee of: Employee of Eli Lilly and Company, Jess Paulus: None declared, Aubrey Trevelin Sprabery Shareholder of: Shareholder of Eli Lilly and Company, Employee of: Employee of Eli Lilly and Company, Ennio Lubrano: None declared