

Background: Despite the availability of effective treatments, up to 10-20% of patients with Rheumatoid Arthritis (RA) do not respond to multiple targeted medications and can be classified as multi-drug resistant (MDR). MDR-RA has been linked to both extrinsic factors, such as comorbidities and non-compliance and, intrinsic factors related to the molecular pathology of the diseased tissue. For example, recently, a stromal synovial signature has been associated to multi-drug resistance. However, the role of immune cells and pathways in mediating multi-drug resistance is unknown.
Objectives: To investigate the contribution of specific synovial immune cells in the pathogenesis of MDR-RA.
Methods: In The R4RA trial, TNF-inhibitors inadequate responders RA patients were randomised to Tocilizumab or Rituximab following a synovial biopsy. At week 16, CDAI50 non-responders were switched to the alternative medication. At the end of the trial, we identified patients who did not respond to at least 3x bDMARDs, defined as MDR-RA (n=39), and compared with first-line (16 weeks) responders to either Rituximab or Tocilizumab (n=65), for baseline demographics, clinical characteristics and synovitis assessed by immunohistochemistry. In silico deconvolution (xCell) and differentially expressed gene (DEG) analyses were used to identify cell types and molecular signatures associated with treatment response/refractoriness specific to the lympho-myeloid population, i.e. with synovial infiltration of B and T cells.
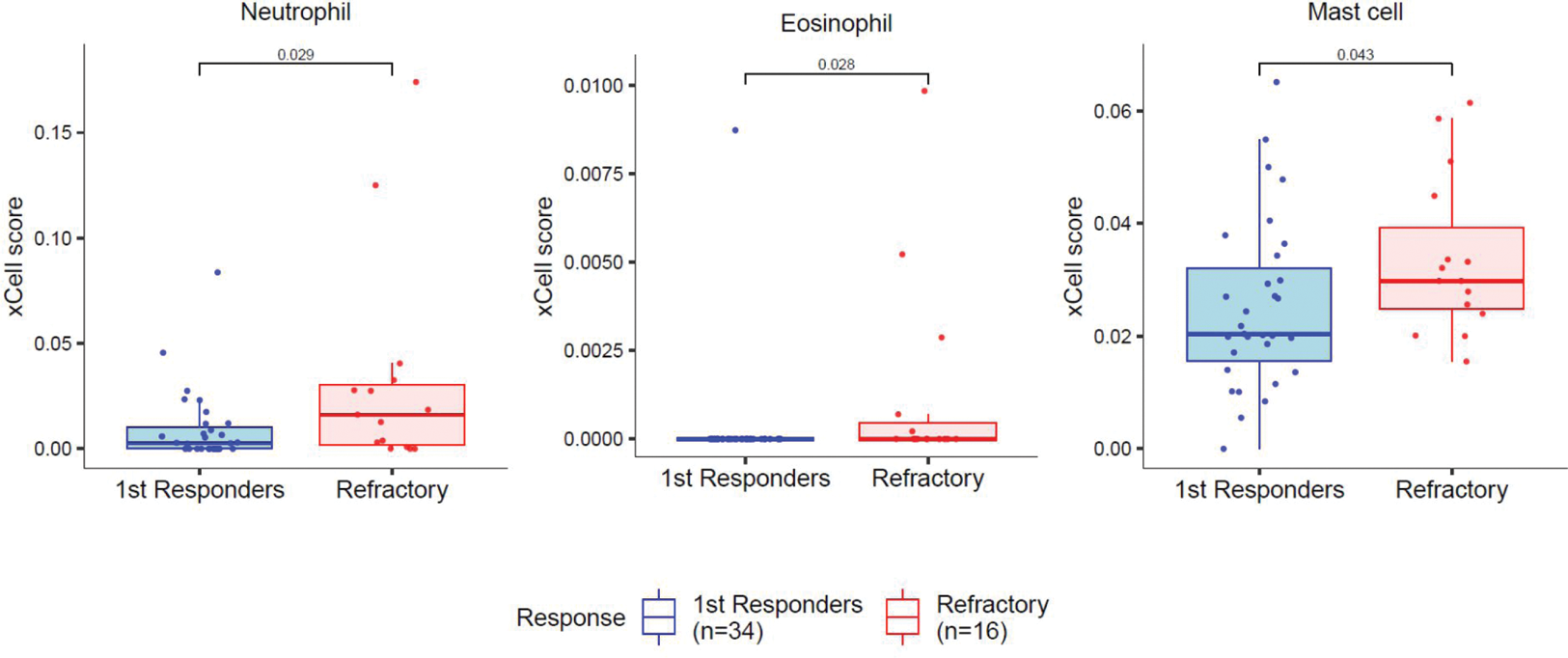
Results: The synovial pathotype distribution was similar in MDR-RA patients and first-line responders, with a comparable prevalence of the lympho-myeloid pathotype, characterised by an abundance of B and T cells—specifically 16 (41%) in MDR-RA and 34 (52.3%) in first-line responders. However, within this highly-inflamed lympho-myeloid population, MDR-RA patients showed significantly higher disease activity (CDAI 37.0 [33.9-41] vs 25.5 [17.6-35.3]; p=0.02), tender joint count (16.0 [12.3-22.3] vs 9.5 [4.0-15.5]; p=0.02), circulating white blood cells (10.3 [7.8-12.3] vs 8.2 [6.7-9.1]; p=0.02) and neutrophils (7.2 [5.6-9.3] vs 5.2 [4.4-6.5]; p=0.004) (Table 1). Molecular analysis showed 39 Differentially Expressed Genes in the MDR-RA lympho-myeloid population, particularly genes related to the innate immunity, such as CXCR1, CXCR2, S100A12, MS4A3, and TNFRSF10C. Accordingly, pathway analysis highlighted pathways related to granulocyte chemotaxis and migration upregulated in MDRA-RA lympho-myeloid patients. In silico deconvolution using xCell showed significantly higher levels of neutrophils, eosinophils, and mast cells in MDR-RA lympho-myeloid patients (Figure 1).
Conclusion: Our study highlights a considerable heterogeneity in synovial inflammation among MDR-RA patients, pointing to diverse mechanisms underlying treatment resistance. Particularly within the lympho-myeloid population, our findings unveil an innate immune signature linked to MDR-RA, suggesting a potential role for innate immune cells in driving treatment resistance through potential crosstalk with adaptive immunity.
REFERENCES: [1] Rivellese et al, Nat Med 28, 1256–1268 (2022).
MDR-RA vs First line responders (lympho-myeloid patients)
| MDR-RA
| 1st line Responders
| p -value | |
|---|---|---|---|
| Age | 56.2 (50.7-68.1) | 58.1 (52.3-65.4) | 0.99 |
| Gender (F) | 13 (81.3) | 24 (70.6) | 0.64 |
| BMI | 26.1 (20.4-30.0) | 26.4 (22.6-32.1) | 0.43 |
| WBC | 10.3 (7.8-12.3) | 8.2 (6.7-9.1) | 0.02 |
| Platelets | 335 (254-405) | 290 (241-263) | 0.08 |
| Neutrophils | 7.2 (5.6-9.3) | 5.2 (4.4-6.5) | 0.004 |
| Lymphocytes | 1.6 (1.4-2.3) | 1.7 (1.4-2.4) | 0.97 |
| RA duration | 9.0 (3.8-25.3) | 10.0 (4.5-16.8) | 0.93 |
| TJC 28 | 16.0 (12.3-22.3) | 9.5 (4.0-15.5) | 0.02 |
| SJC 28 | 7.5 (6.0-9.5) | 4.5 (2.0-9.0) | 0.08 |
| ESR | 42.0 (29.0-59.6) | 28.5 (18.0-43.5) | 0.15 |
| CRP | 19.5 (6.7-47.0) | 10.5 (5.6-28.8) | 0.32 |
| VAS Pain | 66.0 (54.3-81.8) | 73.5 (54.0-88.8) | 0.45 |
| Patient Global Assessment | 75.5 (55.5-81.3) | 71 (54.3-85.5) | 0.84 |
| CDAI | 37.0 (33.9-41) | 25.5 (17.6-35.3) | 0.02 |
| DAS28 CRP | 5.91 (5.26-6.45) | 5.25 (4.16-6.02) | 0.04 |
| DAS28 ESR | 6.6 (5.9-6.8) | 5.4 (4.4-6.5) | 0.04 |
Data shown as median (IQR) or n (% )
Acknowledgements: We thank all patients participating in the trial and the Precision Medicine Patient Advisory Group (PM-PAG) for their continuous support. The R4RA trial was funded by the Efficacy and Mechanism Evaluation (EME) Programme, a partnership between the Medical Research Council (MRC) and the National Institute for Health and Care Research (NIHR) (grant no. 11/100/76). This study was further supported by NIHR (grant 131575) and MRC (MR/V012509/1) and acknowledges the support of the National Institute for Health Research Barts Biomedical Research Centre (NIHR203330).
Disclosure of Interests: None declared.