

Background: Anti-synthetase syndrome is characterized by interstitial lung disease (ILD), myositis, skin rash, and arthritis, in addition to the presence of anti-aminoacyl-tRNA synthetase (ARS) antibodies. Although most patients with anti-synthetase syndrome respond well to immunosuppressive therapy, which includes glucocorticoids (GCs) and immunosuppressants such as calcineurin inhibitors (CNIs), approximately half of the patients experience a flare of the disease when the GC dose is tapered. However, the risk factors for flares in anti-synthetase syndrome have not been fully elucidated.
Objectives: This study aims to clinically characterize the flares of anti-synthetase syndrome and to identify their risk factors.
Methods: We conducted a retrospective cohort study that included consecutive patients who fulfilled the criteria for anti-synthetase syndrome proposed by Solomon et al. and received induction therapy from November 2006 to March 2022. We reviewed medical records and collected data on demographics, disease characteristics, autoantibodies, treatment, and clinical outcomes. Patients were considered to have had a flare when treatment was intensified for myositis and/or ILD with an increased dose of GC and/or addition of a new immunosuppressant. Risk factors for flares were identified by univariate (log-rank test) and multivariate (Cox regression model) analyses.
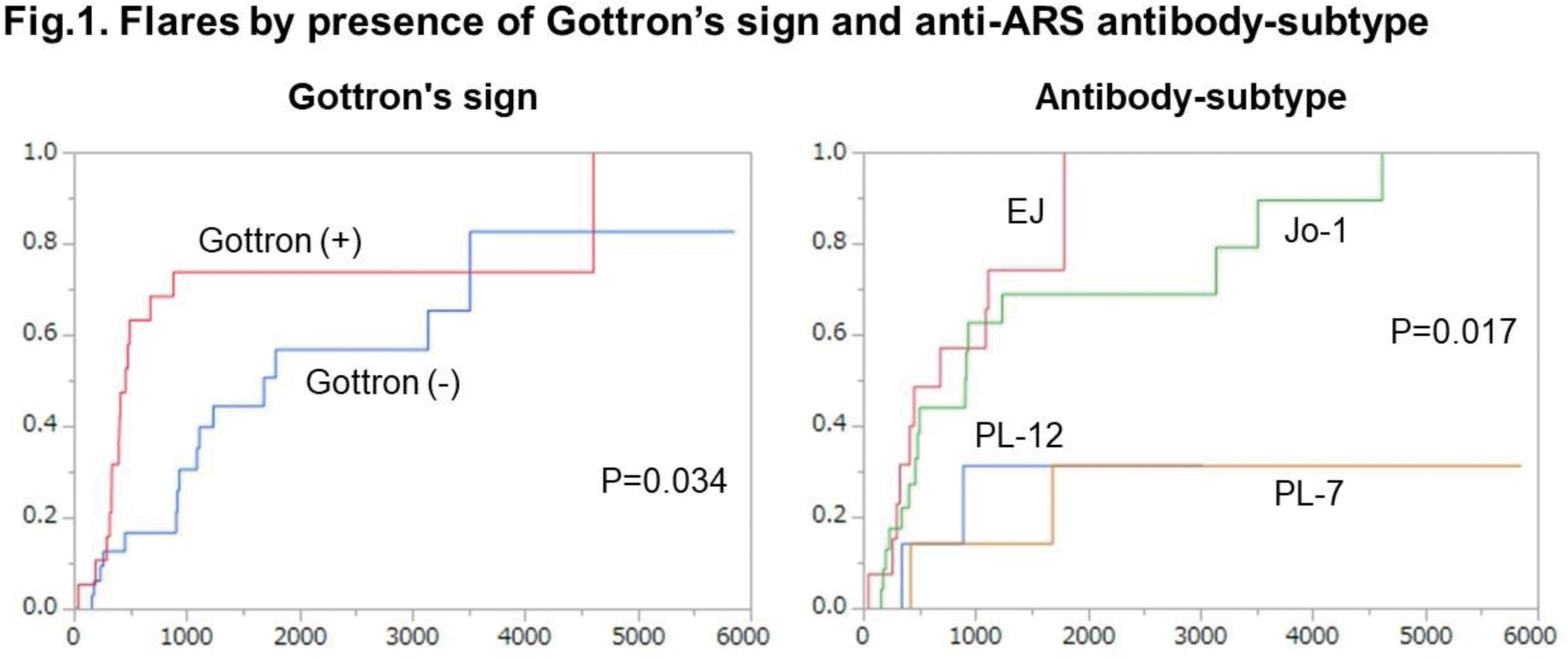
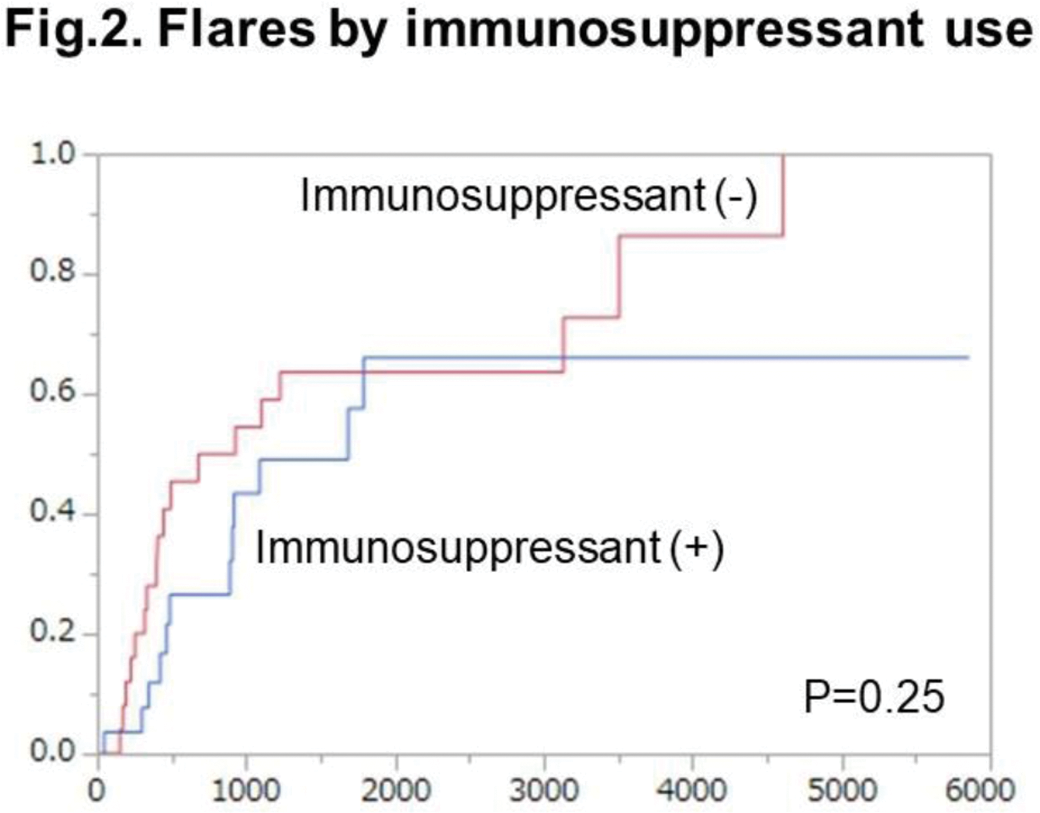
Results: This study included 51 patients, 35 of whom were female, with mean age of 60 years. ILD was found in all patients, with 38 and 39 patients having myositis and skin rash, respectively. Subtypes of anti-synthetase antibodies were anti-Jo-1 antibody in 23 cases (45%), anti-EJ antibody in 13 (25%), anti-PL-7 antibody in 8 (16%), and anti-PL-12 antibody in 7 (14%). As an initial therapy, half of the patients were treated with GC alone, while the rest were treated with a combination of GC and immunosuppressants, many of which were CNIs. Flares occurred in 30 cases (58%) at a median time of 155 weeks. At the time of flare, ILD, myositis, and skin rash developed in 21 (70%), 9 (30%), and 5 (17%) cases, respectively. Flares frequently occurred when the prednisolone dose was tapered to 11 mg/day. In univariate analyses for baseline characteristics, Gottron’s sign, mechanic’s hand, and subtypes of ARS antibodies were identified as significant risk factors for flares, whereas sex, age, and organ involvement were not. Patients with anti-EJ or anti-Jo-1 antibodies more frequently developed flares than those with anti-PL-12 or PL-7 antibodies (Figure 1). In multivariate analyses, anti-ARS subtype and Gottron’s sign were identified as independent risk factors. In univariate analysis for treatment, relapse was not different between patients treated with and without immunosuppressants (P=0.25) (Figure 2). In sub-analyses, the use of immunosuppressants was significantly associated with the relapse of myositis (P=0.03) but not with that of ILD (P=0.95).
Conclusion: Approximately 60% of anti-synthetase syndrome patients experienced a flare, which frequently occurred when the GC dose was tapered to 10 mg of prednisolone. The presence of anti-EJ/Jo-1 antibody and Gottron’s sign were identified as risk factors for flares. The use of immunosuppressants was not identified as a significant preventive factor for flares of anti-synthetase syndrome as a whole, but may have prevented the relapse of myositis.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Anna Hasegawa: None declared, Kazuhiro Kurasawa: None declared, Azusa Kikuchi: None declared, Sara Komatsu: None declared, Yuki Aizawa: None declared, Aya Shimizu: None declared, Yuhi Yoshida: None declared, Tomoka Hiyama: None declared, Tomoyuki Miyao: None declared, Ayae Tanaka: None declared, Satoko Arai: None declared, Reika Maezawa: None declared, Masafumi Arima: None declared, Kei Ikeda Eli Lilly, Abbvie, Eisai, Gilead, AstraZeneca, Asahi Kasei
Grant/research support: Mitsubishi-Tanabe.