

Background: Lymphocytic infiltration of the salivary glands (SGs) represents one hallmark of the autoimmune conditions Sjögren’s Disease (SjD). It is becoming apparent however that loss of SG function precedes presence of immune infiltration, and that other mechanisms must play a role.
Objectives: We sought to elucidate the earliest events in of SG dysfunction in SjD.
Methods: Parotid gland biopsies were harvested from 12 SSA + SjD patients grouped into those with no infiltration (focus score (FS) =0, n =4), minimal infiltration (FS 0-1, n =4) and a positive focus score (FS>1, n =4). Healthy control (HC) parotid glands were also harvested ( n =4). Single cell RNA sequencing was performed on all cells present in all biopsies. Electron microscopy on SG tissue was performed using standard techniques. Peroxidase-based immunohistochemistry with objective quantification using QuPath software was performed, and 5-plex immunoflourescence using the Opal staining kit. Differentiated SG organoids were generated from healthy SGs using our previously published protocol, exposed to GZMK and analyzed using bulk RNASeq.
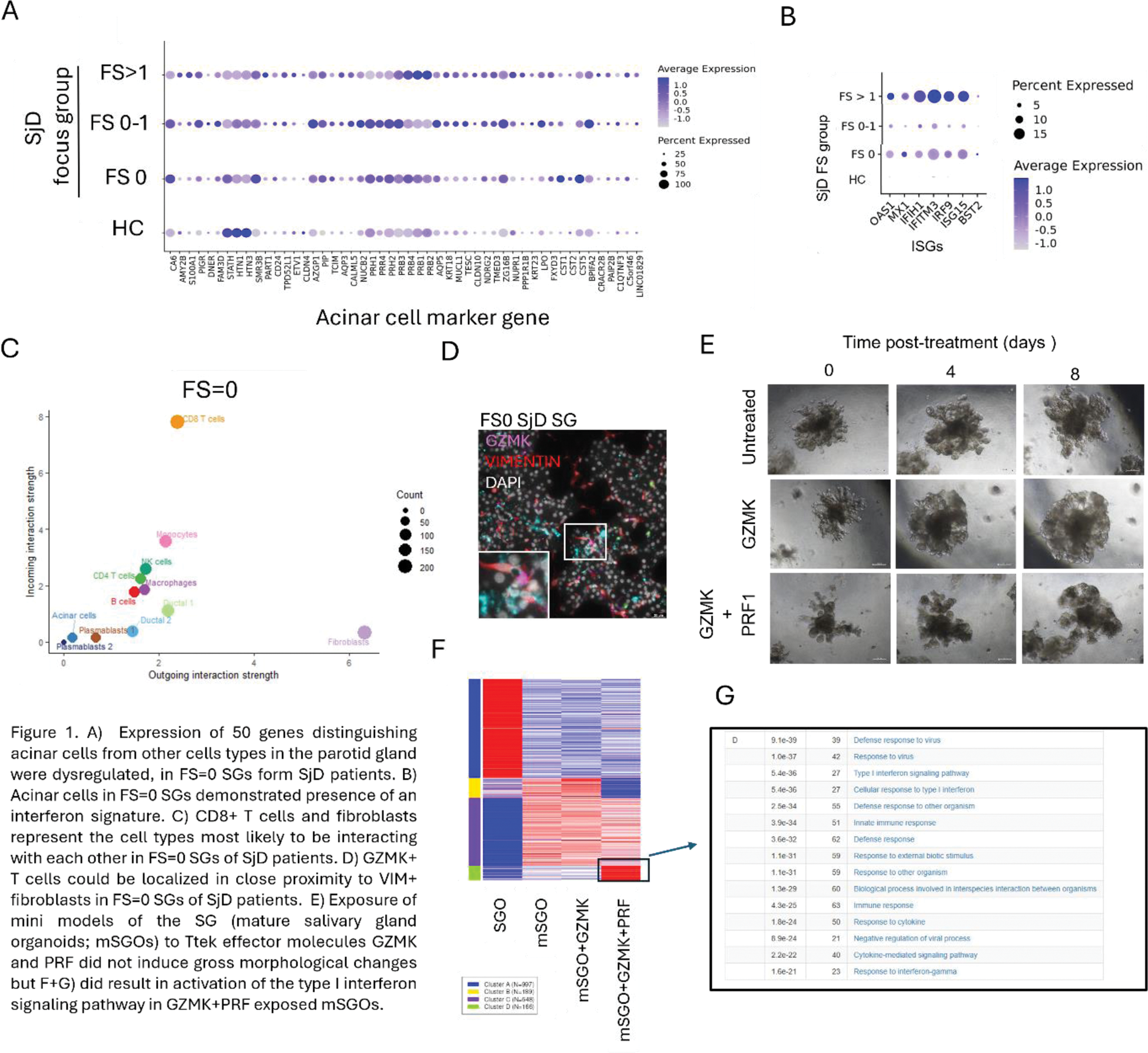
Results: In SGs from SjD patients with FS=0, we observed dysregulation of a panel of 50 genes associated with acinar cell functionality (Figure 1A). Upregulation of 3 genes (PRH2, NKCC1 and PIP) was validated at protein level. Interferon signature was observed in the acinar cells of FS=0 SjD patients (Figure 1B). Mitochondria in acinar cells of FS=0 SjD patients were fewer in number but larger in size compared to HCs. Cellchat analysis of our scRNASeq data suggested that the most likely cell types interacting with each other in FS=0 SGs were fibroblasts as signal producing cells, and CD8 + CD69 + Granzyme K+ (GZMK) T cells (‘Ttek’ cells) as signal receiving cells (Figure 1C). This interaction was predicted to occur via CXCL12 and extracellular matrix (ECM) components produced by fibroblasts. In tissue of FS=0 SjD patients, CD69 + GZMK + Tteks, fibroblasts and acinar cells could be located in close proximity to each other (Figure 1D). SG organoids (SGOs) derived from SG progenitor cells can be differentiated into mature SGOs (mSGOs) containing functional acinar cells (Figure 1E). Exposure of mSGOs generated from HC SGs to GZMK induced dysregulation of acinar cell proteins and mitochondria and initiation of an interferon signature (Figure 1, F and G).
Conclusion: We hypothesize that activated fibroblasts, in the SGs of patients with SjD before presence of lymphocytic infiltration, promotes activation of tissue resident Ttek cells. The Ttek effector molecule GZMK may then trigger acinar cell dysfunction, and represent the very first stages of SG pathology in SjD.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (