

Background: Radiographic progression is a major outcome in axial spondyloarthritis (axSpA), contributing to functional limitations and impaired mobility.
Objectives: Investigating the performance of machine learning (ML) models in predicting spinal radiographic progression over 2-year intervals in axSpA based on clinical and laboratory data.
Methods: Longitudinal data of patients with axSpA obtained from three independent cohorts (GESPIC, CONSUL, ENRADAS) were analysed. Apart from presence of syndesmophytes and axSpA classification status (radiographic vs non-radiographic), both at baseline, clinical (age, sex, symptom duration, BMI, smoking, ASDAS, IBD, uveitis, psoriasis, peripheral arthritis, TNF intake, NSAID intake) and laboratory (CRP, HLA-B27) data were used to train and test five ML models (logistic regression, support vector machine, random forest, XGboost, gradient boosting) in predicting radiographic progression, which was defined as an increase of ≥2 units in the modified Stoke Ankylosing Spondylitis Spine Score over 2-year intervals. Performance of the ML models was assessed using accuracy, precision, recall and F1 score. The latter takes both precision and recall into account and is often used with imbalanced data. All metrics were calculated using both unweighted and weighted methods. Feature importance analysis was conducted using SHAP values for the model with the highest accuracy.
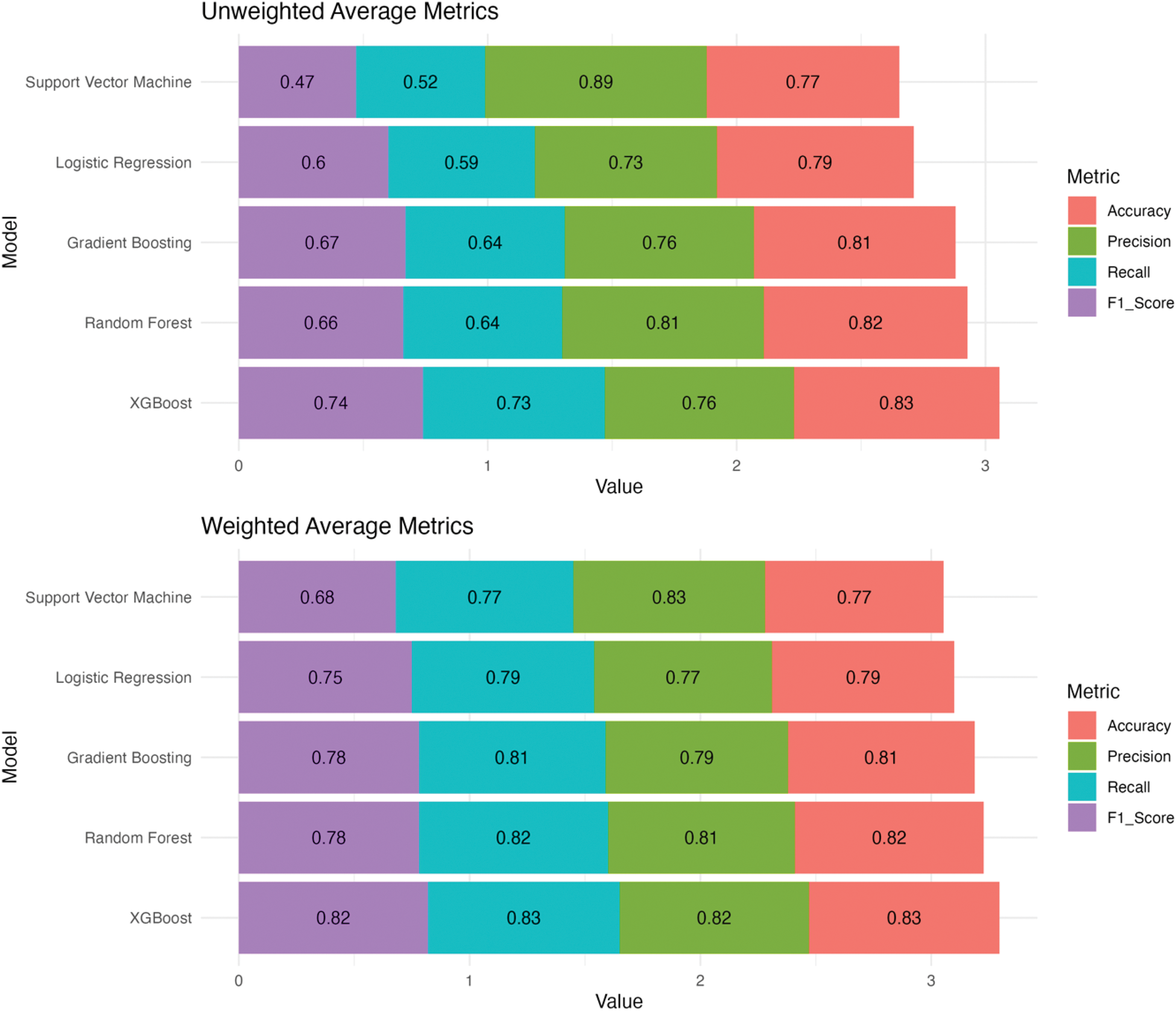
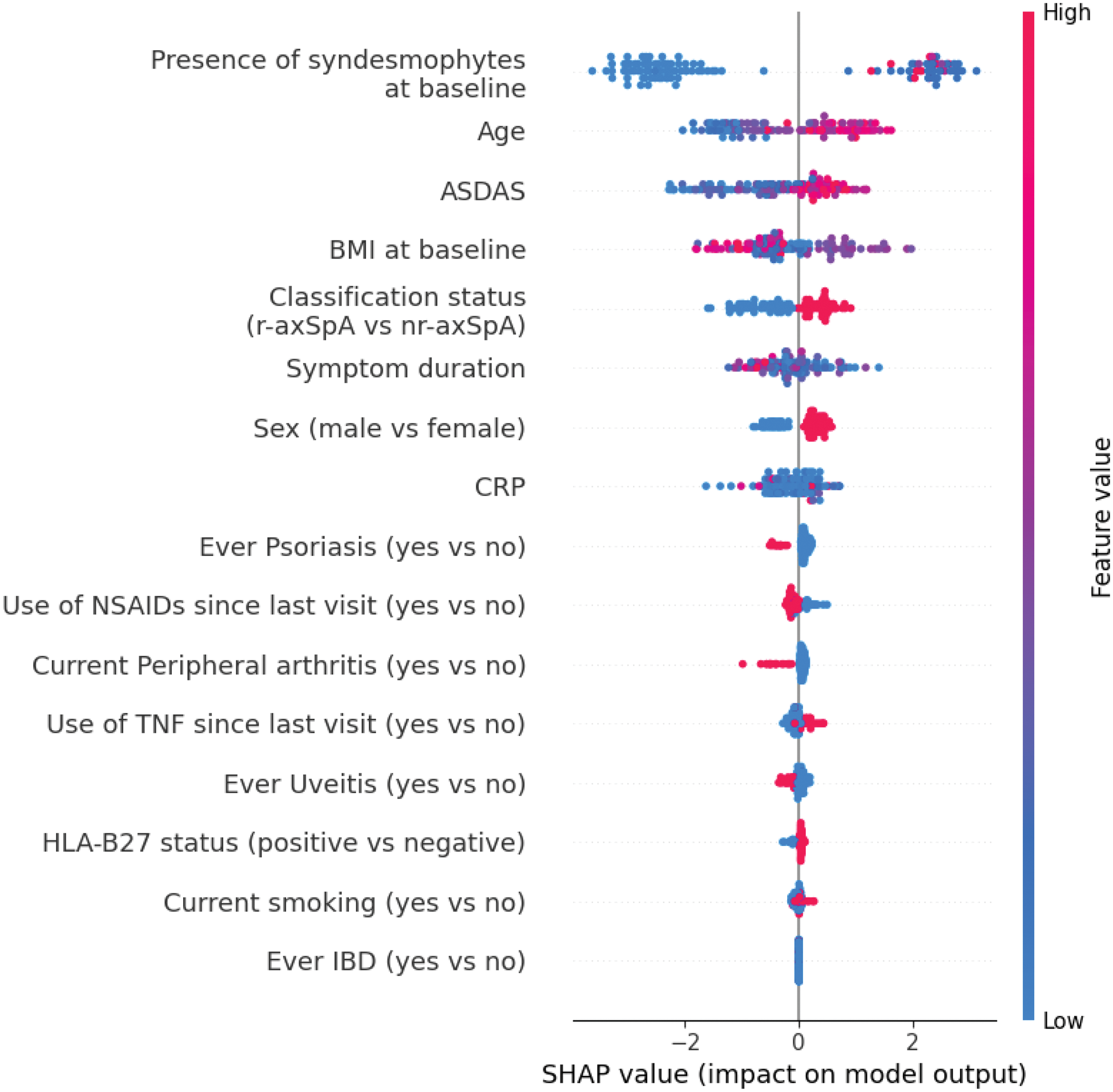
Results: A total of 324 axSpA patients contributing data from 589 follow-up visits, with a mean (SD) follow up of 4.8 (3.1) years, were included. The mean (SD) age at baseline was 39 (10) years and 55% were male. Radiographic progression was observed at 17% of visits among 24% of patients. The unweighted and weighted performance of the ML models is summarized in Figure 1. Among the five models, XGBoost demonstrated the highest accuracy (83%) and F1 score (unweighted: 74%; weighted: 82%), outperforming conventional logistic regression (Figure 1). The contribution of each variable to the prediction of the XGboost model is shown in Figure 2. Presence of syndesmophytes at baseline contributed most, followed by age and ASDAS.
Conclusion: The investigated ML models showed a good overall performance (accuracy) in predicting spinal radiographic progression over 2-year intervals in axSpA patients based on clinical and laboratory data. However, there is potential for improvement, particularly evident in the unweighted metrics, which may be increased by a multi-modal approach including MRIs alongside clinical and laboratory data.
Performance of the machine learning models (unweighted and weighted).
Feature Importance of the XGBoost model calculated by SHAP values.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Imke Redeker: None declared, Murat Torgutalp: None declared, Fabian Proft with payments made directly to FP: AbbVie, AMGEN, BMS, Celgene, Eli Lilly, Hexal, Janssen, Medscape, MSD, Novartis, Pfizer, Roche and UCB, with payments made directly to FP: AbbVie, BMS, Janssen, Novartis, Pfizer and UCB, with payments made via FP’s institution: Novartis, Eli Lilly and UCB, Valeria Rios Rodriguez AbbVie and Takeda, AbbVie, Eli Lily, Jannsen, Pfizer and UCB, Judith Rademacher Janssen and UCB, Mikhail Protopopov Jannsen, Hildrun Haibel UCB, Abbvie, Novartis, Pfizer, Janssen, GSK, Sobi, Abbvie, UCB, Janssen, Sobi, Novartis, Pfizer, Sobi, Novartis, Pfizer, UCB, Alfasigma, Xenofon Baraliakos Abbvie, Alphasigma, Amgen, BMS, Cesas, Celltrion, Galapagos, Janssen, Lilly, Moonlake, Novartis, Pfizer, Roche, Sandoz, Springer, Stada, Takeda, UCB, Zuellig, Abbvie, Alphasigma, Amgen, BMS, Cesas, Celltrion, Galapagos, Janssen, Lilly, Moonlake, Novartis, Pfizer, Roche, Sandoz, Springer, Stada, Takeda, UCB, Zuellig, Abbvie, Alphasigma, Amgen, BMS, Cesas, Celltrion, Galapagos, Janssen, Lilly, Moonlake, Novartis, Pfizer, Roche, Sandoz, Springer, Stada, Takeda, UCB, Zuellig, Abbvie, Celltrion, Janssen, Moonlake, Novartis, Martin Rudwaleit Abbvie, AstraZeneca, Boehringer-Ingelheim, Chugai, Eli Lilly, Janssen, Novartis, UCB, Abbvie, AstraZeneca, Boehringer-Ingelheim, Chugai, Eli Lilly, Janssen, Novartis, UCB, Joachim Sieper Abbvie, Merck, Novartis, Denis Poddubnyy AbbVie, Canon, DKSH, Eli Lilly, Janssen, MSD, Medscape, Novartis, Peervoice, Pfizer, and UCB, AbbVie, Biocad, Bristol-Myers Squibb, Eli Lilly, Janssen, Moonlake, Novartis, Pfizer, and UCB, AbbVie, Eli Lilly, Janssen, Novartis, Pfizer, UCB.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (