

Objectives: To analyse the phenotype of patients with primary Sjogren syndrome (SjS) in whom a lymphoproliferative disease is diagnosed concomitantly.
Methods: By January 2019, The Big Data Sjögren Project included 11,420 consecutive patients with primary SjS recruited from 24 countries of the five continents.
Results: 117 (1%) patients were diagnosed with lymphoma and primary SjS synchronously. Age-gender adjusted multivariate analysis identified the following features associated with lymphoma (OR; CI95%): male gender (4.61; 2.88-7.18), White ethnicity (3.51; 1.78-7.91), abnormal oral tests (3.4; 1.38-10.88), positive biopsy (3.2; 1.3-10.17), positive RF (2.27; 1.48-3.53), hypocomplementemia (3.39; 2.06-5.54), and cryoglobulins (4.74; 2.57-8.38). Activity (score > 1) in the constitutional (2.97; 1.86-4.62), glandular (3.11; 2.1-4.57), cutaneous (2.17; 1.28-3.52), peripheral nerve (2.56; 1.4-4.41) and hematological (2.49; 1.64-3.75) ESSDAI domains was associated with lymphoma (frequencies summarized in the Figure).
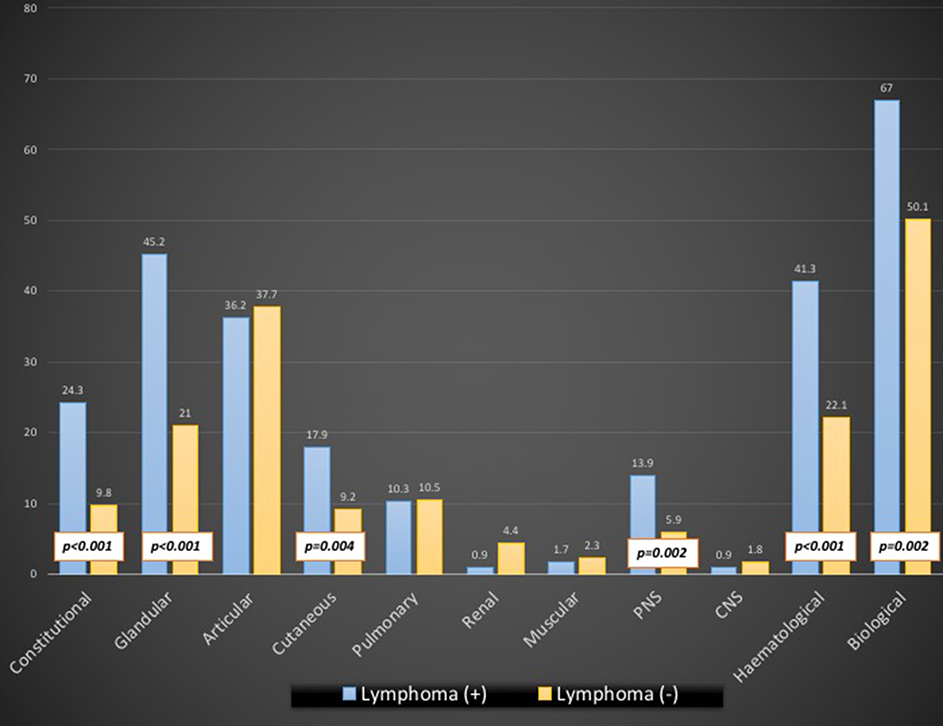
Conclusion: Patients diagnosed concomitantly with primary SjS and lymphoma have a very specific, highly-active phenotype (men, White, severe oral involvement, cryoglobulinemic-related immunological markers, and high systemic activity).
Disclosure of Interests: Soledad Retamozo: None declared, Nihan Acar-Denizli: None declared, Wan Fai Ng: None declared, Antónia Szántó: None declared, Astrid Rasmussen: None declared, Raphaèle Seror Grant/research support from: Pfizer, Consultant for: Bristol-Myers Squibb, Pfizer, Amgen, Eli Lilly, Roche, Celgene, GlaxoSmithKline, MedImmune, Xiaomei Li: None declared, Chiara Baldini: None declared, Jacques-Eric Gottenberg Grant/research support from: Bristol-Myers Squibb, Grant/research support from: Bristol-Myers Squibb, Consultant for: Bristol-Myers Squibb, Lilly, Pfizer, Sanofi-Genzyme, UCB Pharma, Consultant for: Bristol-Myers Squibb, Eli Lilly, UCB, Sanofi-Genzyme, Pfizer, Pulukool Sandhya: None declared, Luca Quartuccio: None declared, Roberta Priori: None declared, Gabriela Hernandez-Molina: None declared, Berkan Armagan: None declared, Aike A. Kruize: None declared, Seung-Ki Kwok: None declared, Marika Kvarnstrom: None declared, Sonja Praprotnik: None declared, Damien Sene: None declared, Roser Solans-Laqué: None declared, Maureen Rischmueller Consultant for: Abbvie, Bristol-Meyer-Squibb, Celgene, Glaxo Smith Kline, Hospira, Janssen Cilag, MSD, Novartis, Pfizer, Roche, Sanofi, UCB, Thomas Mandl: None declared, Yasunori Suzuki: None declared, David Isenberg: None declared, Valeria Valim: None declared, Agata Sebastian: None declared, Gunnel Nordmark: None declared, Hendrika Bootsma: None declared, Hideki Nakamura: None declared, Roberto Giacomelli Grant/research support from: Pfizer, Actelion, Speakers bureau: Actelion, Bristol-Myers Squibb, Merck Sharp & Dohme, Abbvie, Pfizer, Sobi, Roche, Valerie Devauchelle-Pensec Grant/research support from: Roche-Chugai, Speakers bureau: MSD, BMS, UCB, Roche, Benedikt Hofauer Consultant for: Consultant for Galvani Bioelectronics for the area of sleep disorders., Michele Bombardieri Grant/research support from: Celgene, Consultant for: Medimmune, Virginia Fernandes Moça Trevisani: None declared, Daniel Hammenfors: None declared, Sandra Pasoto: None declared, Tamer A Gheita: None declared, Fabiola Atzeni: None declared, Jacques Morel: None declared, Cristina Vollenveider: None declared, Sandra Consani-Fernández: None declared, Xavier Mariette Grant/research support from: Servier, Consultant for: AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Janssen, Pfizer, UCB Pharma, Manuel Ramos-Casals: None declared, Pilar Brito-Zerón: None declared, Elena Bartoloni Bocci: None declared
DOI: 10.1136/annrheumdis-2019-eular.2591