

Background: T cells play a major pathogenic role in Rheumatoid Arthritis (RA). Their pro-inflammatory potential, interaction with B-cells (CD4) and cytotoxic capacities (CD8) have a high impact on disease progression, at the site of inflammation as well as in the periphery. T cells in RA have an altered glucose metabolism, which further amplifies their pro-inflammatory and auto-immune profile. 1, 2 This phenotype is even displayed by RA naïve CD8 T cells, and several studies propose that defects in RA T cell development occur already during thymic development. Thus, acquisition of an activated (auto)inflammatory phenotype may occur at very early stages of RA T cell development.
Objectives: We studied Recent Thymic Emigrants (RTEs) in peripheral blood to establish, whether the activated pro-inflammatory signature present in naïve RA T cells can already be found at such an early stage of T cell development.
Methods: We analysed previously described RTE surface markers 3 on CD8 and CD4 T-cells of human adult thymus samples, as well as on cord-blood samples and additionally confirmed the identified cells as RTEs based on their TREC (T cell Receptor Excision Circle) content. Next, we phenotypically characterised RTEs in the peripheral blood of 10 RA patients, 6 PsA (Psoriatic Arthritis) patients and 5 healthy donors (HC) by 24-parameter Flow Cytometry. Cell clustering was analysed using the UMAP algorithms.
Ethic approval NR: S-969/2020
Results: As expected, RTEs only made up a small proportion of the total CD4 and CD8 T cells, on average 1.3 % and 2.5 % respectively. Contrary to previous reports
4
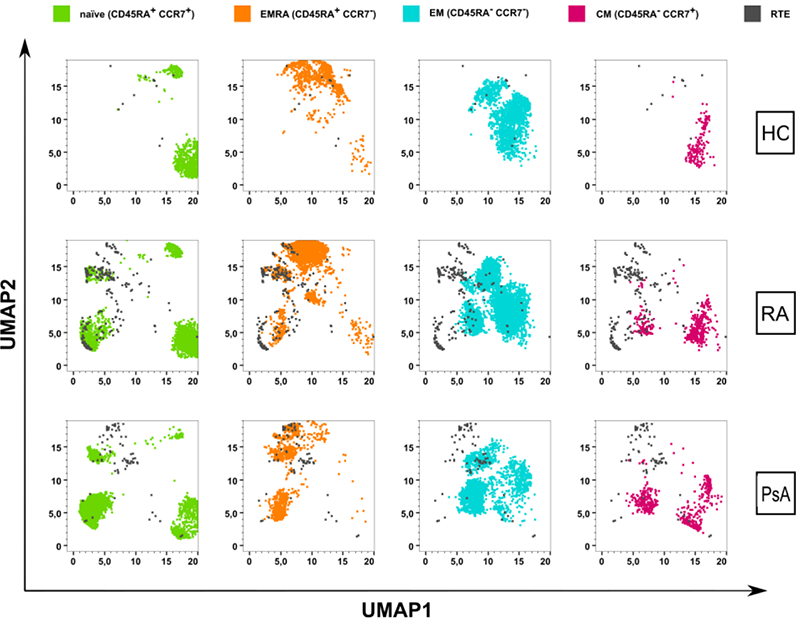
, we could see no significant differences between patients and controls. A first global analysis of CD45RA and CCR7 on CD4 and CD8 via UMAP revealed that RA and PsA RTEs cluster together with naїve and effector cells (
UMAP analysis of total RTEs. Previously downsampled CD4 + and CD8 + T cells were clustered by UMAP (FlowJo™) and total RTEs were mapped on the naїve, effector (EMRA), effector memory (EM) and central memory (CM) compartments of the respective groups.
We further analysed several functional surface markers for homing (CD11a, CD49a), effector functions (CD69, CD27, CD107a), regulatory functions (CD25) and exhaustion (CD95, PD-1, CTLA-4.
In CD4 RTEs we observed more cells expressing homing, effector and exhaustion markers in RA and PsA when compared to HC. CD11a and CD69 were expressed on 3 times more CD4 RTEs in RA and PsA and CD95 and PD1 expressing CD4 RTEs were 2 times more frequent in the arthritic groups compared to HC, while PD1 expressing CD4 RTEs in patients were 10 times more frequent. The CD25 + CD4 RTE population was 3 times larger in RA and PsA than in HC.
In CD8 RTEs the frequency of CD11a expression was similar, whereas the CD49a + population was 3 times larger in RA compared to PsA and HC. CD69 expressing CD8 RTEs were increased in RA and PsA compared to HC, while the degranulation marker CD107a was expressed on similar amounts of HC and PsA RTEs and 2 fold increased compared to RA. CTLA-4 and PD1 expressing CD8 RTEs were 5 - and 2 times more frequent in the RA group than in HC.
Conclusion: Overall, the RTE populations of RA and PsA patients displayed a more active and exhausted phenotype than the control group. However, this seemed to be more prominent for CD4 RTEs than CD8 RTEs. According their CD45RA/CCR7 profiles, both CD4 and CD8 RTEs fall into the less active naїve compartment, corresponding to the early developmental stage, but also into the effector compartment. The RTEs from RA and PsA patients also express markers that are usually associated with pro-inflammatory activity and even exhaustion. This implies that indeed even RTEs are already biased towards a more active phenotype in RA and PsA, compared to RTEs from healthy individuals, suggesting alterations in T cell development in the RA and PsA thymus.
REFERENCES:
[1]
[2]
[3]
[4]
Acknowledgements: We thank all the donors involved in the study for their participation. Furthermore, we thank Christa Stolp and Anke Diehlmann for their indispensable help with the Thymus and Cord Blood samples.
Disclosure of Interests: None declared