

Background Filgotinib (FIL) is a Janus kinase (JAK) 1 preferential inhibitor, approved for treatment of moderate to severe active RA in Europe, the UK, and Japan. Graded laboratory abnormalities from placebo-controlled analyses and long-term data on lymphocytes have been reported previously.
Objectives Report the effect of FIL on laboratory parameters in the FINCH 4 long-term extension (LTE).
Methods Safety was assessed from LTE baseline (BL) to data cutoff (1 Jun 2020) in patients (pts) receiving ≥1 FIL dose (FIL 200 mg [FIL200] or 100 mg [FIL100]) in FINCH 4 (NCT03025308). Laboratory abnormalities were graded per Common Terminology Criteria for Adverse Events v4.03. Frequencies and exposure-adjusted incidence rates (EAIRs)/100 pt-years of exposure (PYE) for graded abnormalities are reported. Median laboratory parameters are reported to LTE Week (W) 48.
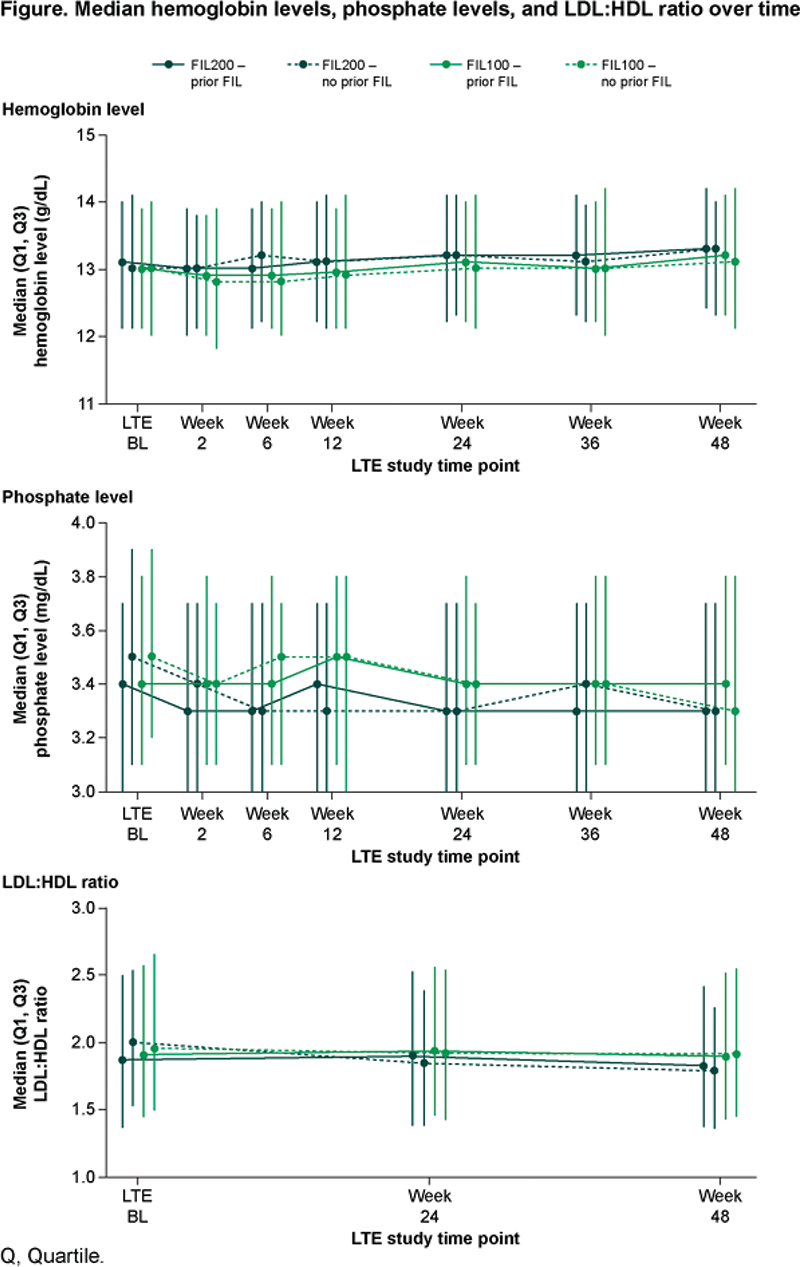
Results In FINCH 4, 2729 pts received FIL for 4198.08 PYE (mean 80.3 weeks); exposure was similar between dose groups. Frequency and EAIR of laboratory abnormalities were similar for anemia, decreased platelets, and increased alanine aminotransferase (ALT), aspartate aminotransferase (AST), and serum creatinine, and higher in the FIL200 vs FIL100 group for neutropenia, increased creatinine kinase (CK), hypophosphatemia, and cholesterol (high) (Table 1; graded data not shown). No Grade 4 decreased phosphate laboratory abnormalities were observed and hypophosphatemia was not associated with adverse events. Laboratory abnormalities led to discontinuation of FIL in 7 pts: ALT and AST increased, 3 (0.2%, FIL 200); ALT increased, 1 (<0.1%, FIL 100); and neutropenia 3 (0.3%, FIL 100). From LTE BL to W48, hemoglobin, platelets, ALT, and AST were relatively stable, with no clear differences between doses, or between pts with or without prior FIL. Neutrophil count was relatively stable from LTE BL to W48. Neutrophils decreased for the FIL200 group with no prior FIL exposure, remaining stable from W24. CK and serum creatinine were relatively stable from LTE BL to W48 in pts with prior FIL exposure; in pts with no prior FIL, initial increases plateaued by W6 and W12, respectively, remaining stable. Changes in phosphate levels from LTE BL were small, remaining within normal range (2.2–5.1 mg/dL). Triglycerides were stable over time with no differences between groups. Total cholesterol, high-density lipoprotein (HDL), and low-density lipoprotein (LDL) levels were stable in pts with prior FIL exposure. In pts with no prior FIL exposure, small increases in total cholesterol, HDL, and LDL plateaued by W24, remaining stable. The LDL:HDL ratio was stable, with no differences between groups (Figure 1).
Conclusion Laboratory abnormalities were generally mild to moderate, similar to previous observations. Frequency and EAIR were higher in the FIL200 vs FIL100 group for neutropenia, increased CK, hypophosphatemia, and high cholesterol.
| n/N (%); EAIR (95% confidence interval) | FIL200 (n=1530; PYE=2346.0) | FIL100 (n=1199; PYE=1852.1) |
|---|---|---|
| Anemia | 264/1526 (17.3); 11.3 (10.0, 12.7) | 227/1193 (19.0); 12.3 (10.8, 14.0) |
| Platelets (decreased) | 44/1525 (2.9); 1.9 (1.4, 2.5) | 24/1189 (2.0); 1.3 (0.9, 1.9) |
| Neutropenia | 194/1526 (12.7); 8.3 (7.2, 9.5) | 101/1193 (8.5); 5.5 (4.5, 6.6) |
| ALT (increased) | 349/1526 (22.9); 14.9 (13.4, 16.5) | 287/1193 (24.1); 15.5 (13.8, 17.4) |
| AST (increased) | 338/1526 (22.1); 14.4 (13.0, 16.0) | 273/1193 (22.9); 14.7 (13.1, 16.6) |
| Serum creatinine (increased) | 80/1526 (5.2); 3.4 (2.7, 4.2) | 62/1193 (5.2); 3.3 (2.6, 4.3) |
| Creatine kinase (increased) | 448/1526 (29.4); 19.1 (17.4, 20.9) | 259/1193 (21.7); 14.0 (12.4, 15.8) |
| Hypophosphatemia | 103/1526 (6.7); 4.4 (3.6, 5.3) | 50/1193 (4.2); 2.7 (2.0, 3.6) |
| Hypertriglyceridemia | 307/1497 (20.5); 13.1 (11.7, 14.6) | 268/1164 (23.0); 14.5 (12.8, 16.3) |
| Cholesterol (high) | 125/1497 (8.4); 5.3 (4.5, 6.3) | 73/1164 (6.3); 3.9 (3.1, 5.0) |
*Defined as an increase of ≥1 toxicity grade from BL up to 30 days post last study drug dose; †Pts who received ≥1 dose of study drug.
Image/graph:
Acknowledgements We thank the physicians and patients who participated in these studies. The FINCH studies were co-funded by Gilead Sciences Inc. (Foster City, CA, USA) and Galapagos NV (Mechelen, Belgium). Publication coordination was provided by Fabien Debailleul, PhD, of Galapagos NV. Medical writing support was provided by Stephanie Rippon, MBio (Aspire Scientific, Bollington, UK) and funded by Galapagos NV.
Disclosure of Interests Ennio Giulio Favalli Speakers bureau: AbbVie, BMS, Celltrion, Galapagos, Janssen, Lilly, MSD, Novartis, Pfizer, UCB, Consultant of: AbbVie, BMS, Celltrion, Galapagos, Janssen, Lilly, MSD, Novartis, Pfizer, UCB, Maya H Buch Speakers bureau: AbbVie (paid to host institution), Consultant of: AbbVie, CESAS Medical, Galapagos, Gilead, Pfizer (all paid to host institution), Grant/research support from: Gilead (paid to host institution), James Galloway Speakers bureau: AbbVie, Biogen, Eli Lilly, Galapagos, Gilead, Janssen, Novartis, Pfizer, Roche, UCB, Consultant of: AbbVie, Eli Lilly, Galapagos, Gilead, Janssen, Novartis, Pfizer, Grant/research support from: AstraZeneca, Celgene, Gilead, Janssen, Medicago, Novavax, Pfizer, Arnaud Constantin Speakers bureau: AbbVie, Amgen, Biogen, BMS, Boehringer, Celltrion, Fresenius-Kabi, Galapagos, Janssen, Lilly, Medac, MSD, Novartis, Pfizer, Roche, Sanofi, Sandoz, UCB, Viatris, Consultant of: AbbVie, Boehringer, Celltrion, Galapagos, Janssen, Lilly, Novartis, Pfizer, UCB, Patrick Durez Speakers bureau: AbbVie, Eli Lilly, Galapagos, Paul Van Hoek Consultant of: Galapagos, Aspen, AOP Health, Sanofi-Genzyme, Astellas, Employee of: Schering Plough, MSD, Janssen, Chris Watson Shareholder of: Galapagos, Employee of: Galapagos, Pieter-Jan Stiers Employee of: Galapagos, Vijay Rajendran Shareholder of: Galapagos, Employee of: Galapagos, Katrien Van Beneden Shareholder of: Galapagos, Employee of: Galapagos, Tsutomu Takeuchi Speakers bureau: AbbVie GK, AYUMI, BMS, Chugai, Daiichi Sankyo, Eisai, Eli Lilly Japan, Gilead Sciences, Inc., Janssen Pharm KK, Mitsubishi Tanabe, Pfizer Japan, Sanofi KK, Consultant of: AbbVie GK, Astellas, Chugai, Eli Lilly Japan, Gilead Sciences, Inc., Janssen Pharm KK, Mitsubishi Tanabe, Pfizer Japan, Grant/research support from: Astellas, AYUMI, Chugai, Daiichi Sankyo, Eisai, Eli Lilly Japan, AbbVie GK, Asahi Kasei Pharma, Mitsubishi Tanabe, Nippon Kayaku, UCB Japan, Bernard Combe Speakers bureau: AbbVie, Celltrion, Galapagos, Janssen, Lilly, Pfizer, Roche-Chugai, Consultant of: AbbVie, Galapagos, Gilead, Janssen, Lilly, Novartis, Roche-Chugai.
Keywords: Clinical trials, Rheumatoid arthritis, Disease-modifying drugs (DMARDs)
DOI: 10.1136/annrheumdis-2023-eular.2129