

Background Despite the increasing number of studies on COVID-19 infection in people with rheumatic and musculoskeletal diseases (RMDs), data on Sjögren’s syndrome (SS) is limited.
Objectives To determine the spectrum and severity of pre-vaccine and post-vaccine breakthrough COVID-19 infections (B-INFs) among patients with SS compared to other connective tissue diseases (CTDs), non-rheumatic autoimmune diseases (nrADs), and healthy controls (HC).
Methods Data were collected using the COVID-19 Vaccination in Autoimmune Diseases (COVAD) questionnaire (March–December 2021) and analysed to compare frequencies and identify associations with demographic, disease-specific and vaccine-specific variables (for B-INFs).
Results Among 9462 complete respondents, 356 were patients with SS, of which 129 had primary (p) SS and 227 had SS associated (a) with rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), systemic sclerosis (SSc) or inflammatory myopathies (IM). Patients with SS were compared to 1586 patients with other CTDs (RA, SLE, SSc or IIM)] but without SS, 825 patients with nrADs and 4712 healthy controls (HC). SS patients had mean age of 52.3 years, mean disease duration was 12 years; 94.5% were females, 68% were Caucasian. Overall COVID-19 infection was reported by 14% of patients with SS and 19.2% of HC (odds ratio (OR)=0.7; 95% confidence interval (CI)=0.5-0.9). The majority of cases occurred prior to the first vaccine dose with no differences across groups in the proportion of B-INFs (SS: 21.6%; other CTD: 20%; nrAD: 19.8%; HC: 19.4%). In the SS group, age and disease duration were similar in patients with either pre-vaccine infections or B-INFs. When comparing pSS and aSS, the overall number of COVID-19 infections was comparable, regardless of the timing (before or after vaccination). Over 80% of SS patients were symptomatic and the symptom duration was similar regardless of the timing of infection, however the symptom burden was different, with certain manifestations absent in B-INFs (oral ulcers, nausea/vomiting, skin rashes, loss of smell/taste) (Figure 1). When focusing on pSS and aSS separately, fever was never reported in aSS B-INFs, whereas other symptoms were equally distributed in pre-vaccine infections and B-INFs. When comparing SS to other CTDs and HC, it emerged that during B-INFs, patients with SS had more frequently chest pain (OR=6.5; 95% CI= 1.8-23 vs NC; OR=11.2; 95% CI=1.7-72 vs other CTDs) and headache (OR= 4.6; 95% CI= 1.1-20 vs other CTDs). Hospitalization for B-INF was required by only one patient with SS patient (pSS) who also required oxygen therapy. Conversely 17% of patients with SS (with an equal distribution between pSS and aSS) required hospitalization for pre-vaccine infections. Although the overall hospitalization rate did not differ across groups, patients with SS needed oxygen therapy more frequently than HCs (OR=9; 95% CI=1.04-78) and other CTDs (OR=19.5; 95% CI= 1.8-213). When comparing pSS and aSS, the need for hospitalization and, if hospitalised, the need of oxygen therapy were comparable for infections before vaccination and B-INFs. In pSS, B-INFs mainly occured after BNT162b2 vaccine (37%) and ChAdOx1 nCoV-19 (37%) with few cases after the other vaccines, whereas in aSS B-INFs occurred after either BNT162b2 vaccine (67%) or BBIBP-CorV (33%).
Conclusion Our data demonstrated that B-INfs in patients with SS are less severe than those observed pre-vaccination. This strengthens the rationale for COVID-19 vaccination in patients with SS.
Image/graph:
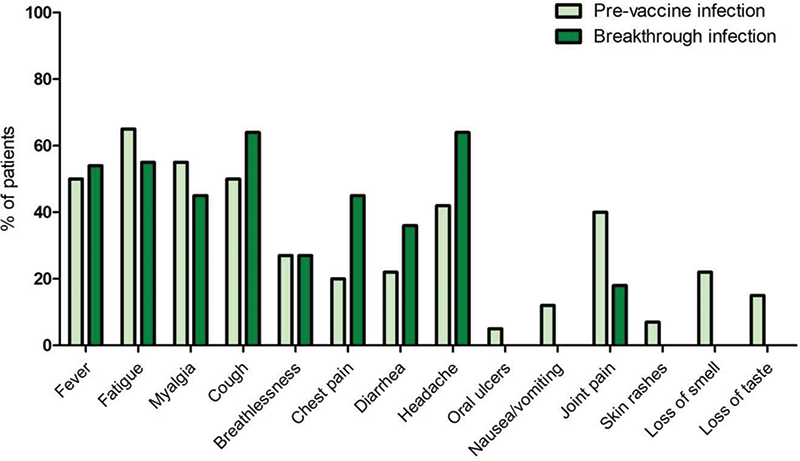
Figure 1. The clinical picture of anti-SARS-CoV-2 infection in patients with Sjögren’s syndrome
Acknowledgements Myositis Assoc., Myositis India, Myositis UK, Myositis Support and Understanding, the Myositis Global Network, Deutsche Gesellschaft für Muskelkranke e.V., Dutch and Swedish Myositis PSG, Cure JM, Cure IBM, Sjögren’s India Found., Patients Engage, Scleroderma India, Lupus UK, Lupus Sweden, Emirates Arthritis Found., EULAR PARE, ArLAR research group, AAAA patient group, Myositis Assoc. of Australia, APLAR myositis SIG, Thai Rheumatism association, PANLAR, AFLAR NRAS, Anti-Synthetase Syndrome SG.
Disclosure of Interests Alessia Alunno: None declared, Francesco Carubbi: None declared, Jessica Day Grant/research support from: CSL Limited, Johannes Knitza: None declared, Samuel Katsuyuki Shinjo: None declared, Sreoshy Saha: None declared, Marcin Milchert: None declared, Oliver Distler Shareholder of: Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143), Speakers bureau: 4P-Pharma, Abbvie, Acceleron, Alcimed, Altavant, Amgen, AnaMar, Arxx, AstraZeneca, Baecon, Blade, Bayer, Boehringer Ingelheim, Corbus, CSL Behring, Galderma, Galapagos, Glenmark, Gossamer, iQvia, Horizon, Inventiva, Janssen, Kymera, Lupin, Medscape, Merck, Miltenyi Biotec, Mitsubishi Tanabe, Novartis, Prometheus, Redxpharma, Roivant, Sanofi and Topadur, Consultant of: 4P-Pharma, Abbvie, Acceleron, Alcimed, Altavant, Amgen, AnaMar, Arxx, AstraZeneca, Baecon, Blade, Bayer, Boehringer Ingelheim, Corbus, CSL Behring, Galderma, Galapagos, Glenmark, Gossamer, iQvia, Horizon, Inventiva, Janssen, Kymera, Lupin, Medscape, Merck, Miltenyi Biotec, Mitsubishi Tanabe, Novartis, Prometheus, Redxpharma, Roivant, Sanofi and Topadur, Grant/research support from: 4P-Pharma, Abbvie, Acceleron, Alcimed, Altavant, Amgen, AnaMar, Arxx, AstraZeneca, Baecon, Blade, Bayer, Boehringer Ingelheim, Corbus, CSL Behring, Galderma, Galapagos, Glenmark, Gossamer, iQvia, Horizon, Inventiva, Janssen, Kymera, Lupin, Medscape, Merck, Miltenyi Biotec, Mitsubishi Tanabe, Novartis, Prometheus, Redxpharma, Roivant, Sanofi and Topadur, Ashima Makol: None declared, Tulika Chatterjee: None declared, Vikas Agarwal: None declared, Rohit Aggarwal Consultant of: Mallinckrodt, Octapharma, CSL Behring, Bristol Myers-Squibb, EMD Serono, Kezar, Pfizer, AstraZeneca, Alexion, Argenx, Boehringer Ingelheim (BI), Corbus, Janssen, Kyverna, Roivant, Merck, Galapagos, Actigraph, Abbvie, Scipher, Horizontal Therapeutics, Teva, Biogen, Beigene, ANI Pharmaceutical, Nuvig, Capella, CabalettaBio, Grant/research support from: Mallinckrodt, Pfizer, Bristol Myers-Squibb, Q32, EMD Serono, Janssen, Boehringer Ingelheim (BI), Latika Gupta: None declared.
Keywords: Vaccination/Immunization, COVID, Sjögren syndrome
DOI: 10.1136/annrheumdis-2023-eular.2499